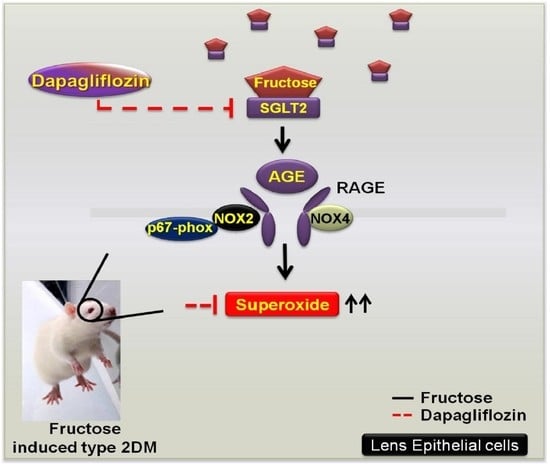
Dapagliflozin Prevents NOX- and SGLT2-Dependent Oxidative Stress in Lens Cells Exposed to Fructose-Induced Diabetes Mellitus
Abstract
:1. Introduction
2. Results
2.1. GLUTs May Act Through RAGE and Induce RAC1 Expression and Superoxide Production in the Cataract Lenses of DM Patients
2.2. Dapagliflozin Prevents Fructose-Mediated Metabolic Defects
2.3. Dapagliflozin Abolishes ROS Production by Inhibiting SGLT2 Expression in LECs of Rats with Fructose-Induced DM
2.4. Dapagliflozin Inhibits GLUT-Induced Expression of NADPH Oxidase Subunits and RAGE in LECs from Rats with Fructose-Induced Type 2 DM
3. Discussion
4. Materials and Methods
4.1. Reagents and Chemicals
4.2. Ethics Statement
4.3. Animals
4.4. Tissue Sample Collection
4.5. Measurement of ROS Production in Lenses from DM Patients and LECs from Type 2 DM Rats
4.6. Measurement of Physiological Indices
4.7. Blood Pressure Measurements
4.8. RNA Isolation and Real-Time Polymerase Chain Reactions (RT-PCR)
4.9. Immunoblotting Analysis
4.10. Immunofluorescence Stainings
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 3-NT | 3-nitrotyrosine |
| AGEs | advanced glycation end products |
| dHDL | direct high-density lipoprotein |
| DM | diabetes mellitus |
| DHE | dihydroethidium |
| DR | diabetic retinopathy |
| GLUT | glucose transporter |
| HbA1c | glycated hemoglobin A1c |
| LECs | lens epithelial cells |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NOX4 | NADPH oxidase 4 |
| RAGE | receptor for advanced glycation end products |
| ROS | reactive oxygen species |
| SGLT2 | sodium-glucose cotransporter 2 |
References
- Tsilas, C.S.; de Souza, R.J.; Mejia, S.B.; Mirrahimi, A.; Cozma, A.I.; Jayalath, V.H.; Ha, V.; Tawfik, R.; Di Buono, M.; Leiter, L.A. Relation of total sugars, fructose and sucrose with incident type 2 diabetes: A systematic review and meta-analysis of prospective cohort studies. CMAJ 2017, 189, E711–E720. [Google Scholar] [CrossRef]
- Maarman, G.J.; Mendham, A.E.; Lamont, K.; George, C. Review of a causal role of fructose-containing sugars in myocardial susceptibility to ischemia/reperfusion injury. Nutr. Res. 2017, 42, 11–19. [Google Scholar] [CrossRef]
- Pollreisz, A.; Schmidt-Erfurth, U. Diabetic cataract-pathogenesis, epidemiology and treatment. J. Ophthalmol. 2010, 2010, 608751. [Google Scholar] [CrossRef]
- Zhang, S.; Chai, F.Y.; Yan, H.; Guo, Y.; Harding, J.J. Effects of N-acetylcysteine and glutathione ethyl ester drops on streptozotocin-induced diabetic cataract in rats. Mol. Vis. 2008, 14, 862–870. [Google Scholar]
- Kumar, P.A.; Reddy, P.Y.; Srinivas, P.N.; Reddy, G.B. Delay of diabetic cataract in rats by the antiglycating potential of cumin through modulation of alpha-crystallin chaperone activity. J. Nutr. Biochem. 2009, 20, 553–562. [Google Scholar] [CrossRef]
- Cheng, P.W.; Chen, Y.Y.; Cheng, W.H.; Lu, P.J.; Chen, H.H.; Chen, B.R.; Yeh, T.C.; Sun, G.C.; Hsiao, M.; Tseng, C.J. Wnt Signaling Regulates Blood Pressure by Downregulating a GSK-3beta-Mediated Pathway to Enhance Insulin Signaling in the Central Nervous System. Diabetes 2015, 64, 3413–3424. [Google Scholar] [CrossRef]
- Francini, F.; Castro, M.C.; Schinella, G.; Garcia, M.E.; Maiztegui, B.; Raschia, M.A.; Gagliardino, J.J.; Massa, M.L. Changes induced by a fructose-rich diet on hepatic metabolism and the antioxidant system. Life Sci. 2010, 86, 965–971. [Google Scholar] [CrossRef]
- Yeh, T.C.; Liu, C.P.; Cheng, W.H.; Chen, B.R.; Lu, P.J.; Cheng, P.W.; Ho, W.Y.; Sun, G.C.; Liou, J.C.; Tseng, C.J. Caffeine intake improves fructose-induced hypertension and insulin resistance by enhancing central insulin signaling. Hypertension 2014, 63, 535–541. [Google Scholar] [CrossRef]
- Bendall, J.K.; Rinze, R.; Adlam, D.; Tatham, A.L.; de Bono, J.; Wilson, N.; Volpi, E.; Channon, K.M. Endothelial Nox2 overexpression potentiates vascular oxidative stress and hemodynamic response to angiotensin II: Studies in endothelial-targeted Nox2 transgenic mice. Circ. Res. 2007, 100, 1016–1025. [Google Scholar] [CrossRef]
- Guglielmotto, M.; Aragno, M.; Tamagno, E.; Vercellinatto, I.; Visentin, S.; Medana, C.; Catalano, M.G.; Smith, M.A.; Perry, G.; Danni, O.; et al. AGEs/RAGE complex upregulates BACE1 via NF-kappaB pathway activation. Neurobiol. Aging 2012, 33, 196-e13. [Google Scholar] [CrossRef]
- Miller, A.; Adeli, K. Dietary fructose and the metabolic syndrome. Curr. Opin. Gastroenterol. 2008, 24, 204–209. [Google Scholar] [CrossRef] [Green Version]
- Wautier, M.P.; Chappey, O.; Corda, S.; Stern, D.M.; Schmidt, A.M.; Wautier, J.L. Activation of NADPH oxidase by AGE links oxidant stress to altered gene expression via RAGE. Am. J. Physiol. Endocrinol. Metab. 2001, 280, E685–E694. [Google Scholar] [CrossRef]
- Wilder-Smith, C.H.; Li, X.; Ho, S.S.; Leong, S.M.; Wong, R.K.; Koay, E.S.; Ferraris, R.P. Fructose transporters GLUT5 and GLUT2 expression in adult patients with fructose intolerance. United Eur. Gastroenterol. J. 2014, 2, 14–21. [Google Scholar] [CrossRef] [Green Version]
- Mantych, G.J.; Hageman, G.S.; Devaskar, S.U. Characterization of glucose transporter isoforms in the adult and developing human eye. Endocrinology 1993, 133, 600–607. [Google Scholar] [CrossRef]
- Nauck, M.A. Update on developments with SGLT2 inhibitors in the management of type 2 diabetes. Drug Des. Dev. Ther. 2014, 8, 1335–1380. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Luo, D.; Zheng, M.; Hao, Y.; Hou, L.; Zhang, S. Effect of pioglitazone on insulin resistance in fructose-drinking rats correlates with AGEs/RAGE inhibition and block of NADPH oxidase and NF kappa B activation. Eur. J. Pharmacol. 2010, 629, 153–158. [Google Scholar] [CrossRef]
- Ma, N.; Siegfried, C.; Kubota, M.; Huang, J.; Liu, Y.; Liu, M.; Dana, B.; Huang, A.; Beebe, D.; Yan, H.; et al. Expression Profiling of Ascorbic Acid-Related Transporters in Human and Mouse Eyes. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3440–3450. [Google Scholar] [CrossRef]
- Berthoud, V.M.; Beyer, E.C. Oxidative stress, lens gap junctions, and cataracts. Antioxid. Redox Signal. 2009, 11, 339–353. [Google Scholar] [CrossRef]
- Nita, M.; Grzybowski, A. The Role of the Reactive Oxygen Species and Oxidative Stress in the Pathomechanism of the Age-Related Ocular Diseases and Other Pathologies of the Anterior and Posterior Eye Segments in Adults. Oxid. Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef]
- Cheng, P.W.; Lin, Y.T.; Ho, W.Y.; Lu, P.J.; Chen, H.H.; Lai, C.C.; Sun, G.C.; Yeh, T.C.; Hsiao, M.; Tseng, C.J.; et al. Fructose induced neurogenic hypertension mediated by overactivation of p38 MAPK to impair insulin signaling transduction caused central insulin resistance. Free Radic. Biol. Med. 2017, 112, 298–307. [Google Scholar] [CrossRef]
- Kojima, M.; Sun, L.; Hata, I.; Sakamoto, Y.; Sasaki, H.; Sasaki, K. Efficacy of alpha-lipoic acid against diabetic cataract in rat. Jpn. J. Ophthalmol. 2007, 51, 10–13. [Google Scholar] [CrossRef]
- Gul, A.; Rahman, M.A.; Hasnain, S.N.; Salim, A.; Simjee, S.U. Could oxidative stress associate with age products in cataractogenesis? Curr. Eye Res. 2008, 33, 669–675. [Google Scholar] [CrossRef]
- Wang, F.; Ma, J.; Han, F.; Guo, X.; Meng, L.; Sun, Y.; Jin, C.; Duan, H.; Li, H.; Peng, Y. DL-3-n-butylphthalide delays the onset and progression of diabetic cataract by inhibiting oxidative stress in rat diabetic model. Sci. Rep. 2016, 6, 19396. [Google Scholar] [CrossRef] [Green Version]
- Lim, J.C.; Perwick, R.D.; Li, B.; Donaldson, P.J. Comparison of the expression and spatial localization of glucose transporters in the rat, bovine and human lens. Exp. Eye Res. 2017, 161, 193–204. [Google Scholar] [CrossRef]
- Shepherd, P.R.; Gibbs, E.M.; Wesslau, C.; Gould, G.W.; Kahn, B.B. Human small intestine facilitative fructose/glucose transporter (GLUT5) is also present in insulin-responsive tissues and brain. Investigation of biochemical characteristics and translocation. Diabetes 1992, 41, 1360–1365. [Google Scholar] [CrossRef]
- Chan, C.Y.; Guggenheim, J.A.; To, C.H. Is active glucose transport present in bovine ciliary body epithelium? Am. J. Physiol. Cell Physiol. 2007, 292, C1087–C1093. [Google Scholar] [CrossRef] [Green Version]
- Hashim, Z.; Zarina, S. Advanced glycation end products in diabetic and non-diabetic human subjects suffering from cataract. Age 2011, 33, 377–384. [Google Scholar] [CrossRef]
- Elliott, S.S.; Keim, N.L.; Stern, J.S.; Teff, K.; Havel, P.J. Fructose, weight gain, and the insulin resistance syndrome. Am. J. Clin. Nutr. 2002, 76, 911–922. [Google Scholar] [CrossRef]
- Leibowitz, A.; Rehman, A.; Paradis, P.; Schiffrin, E.L. Role of T Regulatory Lymphocytes in the Pathogenesis of High-Fructose Diet-Induced Metabolic Syndrome. Hypertension 2013, 61, 1316–1321. [Google Scholar] [CrossRef]
- Conde, S.V.; da Silva, T.N.; Gonzalez, C.; Carmo, M.M.; Monteiro, E.C.; Guarino, M.P. Chronic caffeine intake decreases circulating catecholamines and prevents diet-induced insulin resistance and hypertension in rats. Br. J. Nutr. 2012, 107, 86–95. [Google Scholar] [CrossRef]
- Chao, E.C.; Henry, R.R. SGLT2 inhibition—A novel strategy for diabetes treatment. Nat. Rev. Drug Discov. 2010, 9, 551–559. [Google Scholar] [CrossRef]
- Chylack, L.T., Jr.; Wolfe, J.K.; Singer, D.M.; Leske, M.C.; Bullimore, M.A.; Bailey, I.L.; Friend, J.; McCarthy, D.; Wu, S.Y. The Lens Opacities Classification System III. The Longitudinal Study of Cataract Study Group. Arch. Ophthalmol. 1993, 111, 831–836. [Google Scholar] [CrossRef]
- Kilkenny, C.; Browne, W.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Animal research: Reporting in vivo experiments: The ARRIVE guidelines. Br. J. Pharmacol. 2010, 160, 1577–1579. [Google Scholar] [CrossRef]
- McGrath, J.C.; Lilley, E. Implementing guidelines on reporting research using animals (ARRIVE etc.): New requirements for publication in BJP. Br. J. Pharmacol. 2015, 172, 3189–3193. [Google Scholar] [CrossRef]
- Curtis, M.J.; Bond, R.A.; Spina, D.; Ahluwalia, A.; Alexander, S.P.; Giembycz, M.A.; Gilchrist, A.; Hoyer, D.; Insel, P.A.; Izzo, A.A.; et al. Experimental design and analysis and their reporting: New guidance for publication in BJP. Br. J. Pharmacol. 2015, 172, 3461–3471. [Google Scholar] [CrossRef]





| Control Group (n = 30) | DM Group (n = 30) | p Value | |
|---|---|---|---|
| Age (years) | 62.0 ± 9.5 | 62.3 ± 7.7 | 0.80 |
| Sex (male:female) | 18:12 | 15:15 | 0.34 |
| BMI | 19.2 ± 2.6 | 21.1 ± 2.9 | 0.90 |
| HbA1c | 5.50 ± 0.05 | 7.60 ± 1.02 | 0.01 * |
| Parameter | Control | Fructose 8 W | Fructose 8 W + Dapagliflozin |
|---|---|---|---|
| Systolic blood pressure (mmHg) | 113.2 ± 2.5 | 150.1 ± 2.0 ** | 124.8 ± 1.0 †† |
| Fasting serum fructose (mg/dL) | 2.9 ± 0.2 | 10.51 ± 1.0 ** | 4.5 ± 0.3 †† |
| dHDL (mg/dL) | 50 ± 0.3 | 52.3 ± 0.7 | 50.0 ± 0.6 |
| Triglycerides (mg/dL) | 86.0 ± 0.6 | 256.0 ± 2.3 ** | 220.7 ± 6.1 †† |
| Cholesterol (mg/dL) | 110.8 ± 0.7 | 105.3 ± 2.4 | 103.3 ± 0.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-Y.; Wu, T.-T.; Ho, C.-Y.; Yeh, T.-C.; Sun, G.-C.; Kung, Y.-H.; Wong, T.-Y.; Tseng, C.-J.; Cheng, P.-W. Dapagliflozin Prevents NOX- and SGLT2-Dependent Oxidative Stress in Lens Cells Exposed to Fructose-Induced Diabetes Mellitus. Int. J. Mol. Sci. 2019, 20, 4357. https://doi.org/10.3390/ijms20184357
Chen Y-Y, Wu T-T, Ho C-Y, Yeh T-C, Sun G-C, Kung Y-H, Wong T-Y, Tseng C-J, Cheng P-W. Dapagliflozin Prevents NOX- and SGLT2-Dependent Oxidative Stress in Lens Cells Exposed to Fructose-Induced Diabetes Mellitus. International Journal of Molecular Sciences. 2019; 20(18):4357. https://doi.org/10.3390/ijms20184357
Chicago/Turabian StyleChen, Ying-Ying, Tsung-Tien Wu, Chiu-Yi Ho, Tung-Chen Yeh, Gwo-Ching Sun, Ya-Hsin Kung, Tzyy-Yue Wong, Ching-Jiunn Tseng, and Pei-Wen Cheng. 2019. "Dapagliflozin Prevents NOX- and SGLT2-Dependent Oxidative Stress in Lens Cells Exposed to Fructose-Induced Diabetes Mellitus" International Journal of Molecular Sciences 20, no. 18: 4357. https://doi.org/10.3390/ijms20184357
APA StyleChen, Y. -Y., Wu, T. -T., Ho, C. -Y., Yeh, T. -C., Sun, G. -C., Kung, Y. -H., Wong, T. -Y., Tseng, C. -J., & Cheng, P. -W. (2019). Dapagliflozin Prevents NOX- and SGLT2-Dependent Oxidative Stress in Lens Cells Exposed to Fructose-Induced Diabetes Mellitus. International Journal of Molecular Sciences, 20(18), 4357. https://doi.org/10.3390/ijms20184357