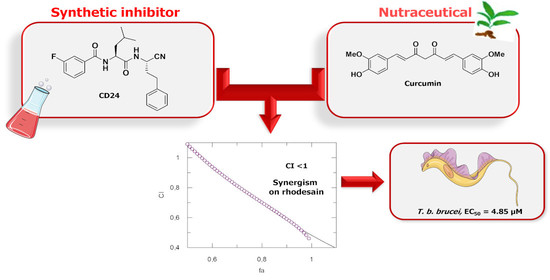
Drug Combination Studies of the Dipeptide Nitrile CD24 with Curcumin: A New Strategy to Synergistically Inhibit Rhodesain of Trypanosoma brucei rhodesiense
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Rhodesain Inhibition Assays
3.2. Antitrypanosomal Activity Assay
3.3. Cytotoxicity Evaluation
3.4. Statistical Analyses
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Human African Trypanosomiasis (Sleeping Sickness). Available online: https://www.who.int/trypanosomiasis_african/en/ (accessed on 4 September 2022).
- Büscher, P.; Cecchi, G.; Jamonneau, V.; Priotto, G. Human African Trypanosomiasis. Lancet 2017, 390, 2397–2409. [Google Scholar] [CrossRef]
- Kansiime, F.; Adibaku, S.; Wamboga, C.; Idi, F.; Kato, C.D.; Yamuah, L.; Vaillant, M.; Kioy, D.; Olliaro, P.; Matovu, E. A multicentre, randomised, non-inferiority clinical trial comparing a nifurtimox-eflornithine combination to standard eflornithine monotherapy for late stage Trypanosoma brucei gambiense human African trypanosomiasis in Uganda. Parasit. Vectors 2018, 11, 105. [Google Scholar] [CrossRef] [PubMed]
- Deeks, E.D. Fexinidazole: First global approval. Drugs 2019, 79, 215–220. [Google Scholar] [CrossRef] [PubMed]
- CHMP Recommends First Oral-Only Treatment for Sleeping Sickness. Available online: https://www.ema.europa.eu/en/news/chmp-recommends-first-oral-only-treatment-sleeping-sickness (accessed on 5 September 2022).
- Navarro, M.; Justo, R.M.S.; Delgado, G.Y.S.; Visbal, G. Metallodrugs for the treatment of trypanosomatid diseases: Recent advances and new insights. Curr. Pharm. Des. 2021, 27, 1763–1789. [Google Scholar] [CrossRef] [PubMed]
- Previti, S.; Di Chio, C.; Ettari, R.; Zappalà, M. Dual inhibition of parasitic targets: A valuable strategy to treat malaria and neglected tropical diseases. Curr. Med. Chem. 2022, 29, 2952–2978. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, M.S.; Hayat, F.; Shin, D. Recent advances in the discovery of novel antiprotozoal agents. Molecules 2019, 24, 3886. [Google Scholar] [CrossRef] [Green Version]
- Kryshchyshyn, A.; Kaminskyy, D.; Grellier, P.; Lesyk, R. Trends in research of antitrypanosomal agents among synthetic heterocycles. Eur. J. Med. Chem. 2014, 85, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Ettari, R.; Previti, S.; Tamborini, L.; Cullia, G.; Grasso, S.; Zappalà, M. The inhibition of cysteine proteases rhodesain and TbCatB: A valuable approach to treat Human African Trypanosomiasis. Mini Rev. Med. Chem. 2016, 16, 1374–1391. [Google Scholar] [CrossRef] [PubMed]
- Ettari, R.; Tamborini, L.; Angelo, I.C.; Micale, N.; Pinto, A.; De Micheli, C.; Conti, P. Inhibition of rhodesain as a novel therapeutic modality for human African trypanosomiasis. J. Med. Chem. 2013, 56, 5637–5658. [Google Scholar] [CrossRef]
- Nikolskaia, O.V.; de Lima, A.A.P.; Kim, Y.V.; Lonsdale-Eccles, J.D.; Fukuma, T.; Scharfstein, J.; Grab, D.J. Blood-brain barrier traversal by African trypanosomes requires calcium signaling induced by parasite cysteine protease. J. Clin. Invest. 2006, 116, 2739–2747. [Google Scholar] [CrossRef]
- Barry, J.D.; McCulloch, R. Antigenic variation in trypanosomes: Enhanced phenotypic variation in a eukaryotic parasite. Adv. Parasitol. 2001, 49, 1–70. [Google Scholar] [CrossRef] [PubMed]
- Lalmanach, G.; Boulange, A.; Serveau, C.; Lecaille, F.; Scharfstein, J.; Gauthier, F.; Authie, E. Congopain from Trypanosoma congolense: Drug target and vaccine candidate. Biol. Chem. 2002, 383, 739–749. [Google Scholar] [CrossRef]
- Ettari, R.; Zappalà, M.; Micale, N.; Schirmeister, T.; Gelhaus, C.; Leippe, M.; Evers, A.; Grasso, S. Synthesis of novel peptidomimetics as inhibitors of protozoan cysteine proteases falcipain-2 and rhodesain. Eur. J. Med. Chem. 2010, 45, 3228–3233. [Google Scholar] [CrossRef] [PubMed]
- Bova, F.; Ettari, R.; Micale, N.; Carnovale, C.; Schirmeister, T.; Gelhaus, C.; Leippe, M.; Grasso, S.; Zappalà, M. Constrained peptidomimetics as antiplasmodial falcipain-2 inhibitors. Bioorg. Med. Chem. 2010, 18, 4928–4938. [Google Scholar] [CrossRef] [PubMed]
- Ettari, R.; Micale, N.; Grazioso, G.; Bova, F.; Schirmeister, T.; Grasso, S.; Zappalà, M. Synthesis and molecular modeling studies of derivatives of a highly potent peptidomimetic vinyl ester as falcipain-2 inhibitors. ChemMedChem 2012, 7, 1594–1600. [Google Scholar] [CrossRef]
- Ettari, R.; Tamborini, L.; Angelo, I.C.; Grasso, S.; Schirmeister, T.; Lo Presti, L.; De Micheli, C.; Pinto, A.; Conti, P. Development of rhodesain inhibitors with a 3-bromoisoxazoline warhead. ChemMedChem 2013, 8, 2070–2076. [Google Scholar] [CrossRef]
- Ettari, R.; Pinto, A.; Tamborini, L.; Angelo, I.C.; Grasso, S.; Zappalà, M.; Capodicasa, N.; Yzeiraj, L.; Gruber, E.; Aminake, M.N.; et al. Synthesis and biological evaluation of papain-family cathepsin L-like cysteine protease inhibitors containing a 1,4-benzodiazepine scaffold as antiprotozoal agents. ChemMedChem 2014, 9, 1817–1825. [Google Scholar] [CrossRef]
- Ettari, R.; Pinto, A.; Previti, S.; Tamborini, L.; Angelo, I.C.; La Pietra, V.; Marinelli, L.; Novellino, E.; Schirmeister, T.; Zappalà, M.; et al. Development of novel dipeptide-like rhodesain inhibitors containing the 3-bromoisoxazoline warhead in a constrained conformation. Bioorg. Med. Chem. 2015, 23, 7053–7060. [Google Scholar] [CrossRef]
- Ettari, R.; Previti, S.; Cosconati, S.; Maiorana, S.; Schirmeister, T.; Grasso, S.; Zappalà, M. Development of novel 1,4-benzodiazepine-based Michael acceptors as antitrypanosomal agents. Bioorg. Med. Chem. Lett. 2016, 26, 3453–3456. [Google Scholar] [CrossRef]
- Ettari, R.; Previti, S.; Cosconati, S.; Kesselring, J.; Schirmeister, T.; Grasso, S.; Zappalà, M. Synthesis and biological evaluation of novel peptidomimetics as rhodesain inhibitors. J. Enzyme Inhib. Med. Chem. 2016, 31, 1184–1191. [Google Scholar] [CrossRef]
- Previti, S.; Ettari, R.; Cosconati, S.; Amendola, G.; Chouchene, K.; Wagner, A.; Hellmich, U.A.; Ulrich, K.; Krauth-Siegel, R.L.; Wich, P.R.; et al. Development of novel peptide-based Michael acceptors targeting rhodesain and falcipain-2 for the treatment of Neglected Tropical Diseases (NTDs). J. Med. Chem. 2017, 60, 6911–6923. [Google Scholar] [CrossRef] [PubMed]
- Ettari, R.; Previti, S.; Maiorana, S.; Amendola, G.; Wagner, A.; Cosconati, S.; Schirmeister, T.; Hellmich, U.A.; Zappalà, M. Optimization strategy of novel peptide-based Michael acceptors for the treatment of Human African Trypanosomiasis. J. Med. Chem. 2019, 62, 10617–10629. [Google Scholar] [CrossRef] [PubMed]
- Maiorana, S.; Ettari, R.; Previti, S.; Amendola, G.; Wagner, A.; Cosconati, S.; Hellmich, U.A.; Schirmeister, T.; Zappala, M. Peptidyl vinyl ketone irreversible inhibitors of rhodesain: Modifications of the P2 fragment. ChemMedChem 2020, 15, 1552–1561. [Google Scholar] [CrossRef]
- Schirmeister, T.; Schmitz, J.; Jung, S.; Schmenger, T.; Krauth-Siegel, R.L.; Gutschow, M. Evaluation of dipeptide nitriles as inhibitors of rhodesain, a major cysteine protease of Trypanosoma brucei. Bioorg. Med. Chem. Lett. 2017, 27, 45–50. [Google Scholar] [CrossRef]
- Giroud, M.; Kuhn, B.; Saint-Auret, S.; Kuratli, C.; Martin, R.E.; Schuler, F.; Diederich, F.; Kaiser, M.; Brun, R.; Schirmeister, T.; et al. 2 H-1,2,3-Triazole-based dipeptidyl nitriles: Potent, selective, and trypanocidal rhodesain inhibitors by structure-based design. J. Med. Chem. 2018, 61, 3370–3388. [Google Scholar] [CrossRef] [PubMed]
- Lemke, C.; Cianni, L.; Feldmann, C.; Gilberg, E.; Yin, J.; Dos Reis Rocho, F.; de Vita, D.; Bartz, U.; Bajorath, J.; Montanari, C.A.; et al. N-Sulfonyl dipeptide nitriles as inhibitors of human cathepsin S: In silico design, synthesis and biochemical characterization. Bioorg. Med. Chem. Lett. 2020, 30, 127420. [Google Scholar] [CrossRef]
- Di Chio, C.; Previti, S.; Amendola, G.; Ravichandran, R.; Wagner, A.; Cosconati, S.; Hellmich, U.A.; Schirmeister, T.; Zappalà, M.; Ettari, R. Development of novel dipeptide nitriles as inhibitors of rhodesain of Trypanosoma brucei rhodesiense. Eur. J. Med. Chem. 2022, 236, 114328. [Google Scholar] [CrossRef]
- Ettari, R.; Previti, S.; Maiorana, S.; Allegra, A.; Schirmeister, T.; Grasso, S.; Zappalà, M. Drug combination studies of curcumin and genistein against rhodesain of Trypanosoma brucei rhodesiense. Nat. Prod. Res. 2019, 33, 3577–3581. [Google Scholar] [CrossRef] [PubMed]
- Ettari, R.; Previti, S.; Di Chio, C.; Maiorana, S.; Allegra, A.; Schirmeister, T.; Zappalà, M. Drug synergism: Studies of combination of RK-52 and curcumin against rhodesain of Trypanosoma brucei rhodesiense. ACS Med. Chem. Lett. 2020, 11, 806–810. [Google Scholar] [CrossRef]
- Di Chio, C.; Previti, S.; De Luca, F.; Allegra, A.; Zappalà, M.; Ettari, R. Drug combination studies of PS-1 and quercetin against rhodesain of Trypanosoma brucei rhodesiense. Nat. Prod. Res. 2022, 36, 4282–4286. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C.; Talalay, P. Quantitative analysis of dose-effect relationships: The combined effects of multiple drugs or enzyme inhibitors. Adv. Enzyme Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef]
- Chou, T.C. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010, 70, 440–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pourkavoos, N. Unique risks, benefits, and challenges of developing drug-drug combination products in a pharmaceutical industrial setting. Comb. Prod. Ther. 2012, 2, 2. [Google Scholar] [CrossRef] [Green Version]
- Previti, S.; Ettari, R.; Calcaterra, E.; Di Chio, C.; Ravichandran, R.; Zimmer, C.; Hammerschmidt, S.; Wagner, A.; Cosconati, S.; Schirmeister, T.; et al. Development of urea bond-containing Michael acceptors as antitrypanosomal agents targeting rhodesain. ACS Med. Chem. Lett. 2022, 13, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.; Bray, M.A.; Cal, M.; Bourdin Trunz, B.; Torreele, E.; Brun, R. Antitrypanosomal activity of fexinidazole, a new oral nitroimidazole drug candidate for treatment of sleeping sickness. Antimicrob. Agents. Chemother. 2011, 55, 5602–5608. [Google Scholar] [CrossRef] [Green Version]
- Caffrey, C.R.; Hansell, E.; Lucas, K.D.; Brinen, L.S.; Alvarez Hernandez, A.; Cheng, J.; Gwaltney, S.L., II; Roush, W.R.; Stierhof, Y.D.; Bogyo, M.; et al. Active site mapping, biochemical properties and subcellular localization of rhodesain, the major cysteine protease of Trypanosoma brucei rhodesiense. Mol. Biochem. Parasitol. 2001, 118, 61–73. [Google Scholar] [CrossRef]
- Biebinger, S.; Elizabeth Wirtz, L.; Lorenz, P.; Christine, C. Vectors for inducible expression of toxic gene products in bloodstream and procyclic Trypanosoma brucei. Mol. Biochem. Parasitol. 1997, 85, 99–112. [Google Scholar] [CrossRef]
- Bogacz, M.; Krauth-Siegel, R.L. Tryparedoxin peroxidase-deficiency commits trypanosomes to ferroptosis-type cell death. eLife 2018, 7, e37503. [Google Scholar] [CrossRef]
- Bogacz, M.; Krauth-Siegel, R. EC50-Determination of RSL3 towards bloodstream T. brucei. Bio-protocol Preprint. 2020. Available online: http://bio-protocol.org/prep654 (accessed on 20 September 2022).
- Previti, S.; Ettari, R.; Di Chio, C.; Ravichandran, R.; Bogacz, M.; Hellmich, U.A.; Schirmeister, T.; Cosconati, S.; Zappala, M. Development of reduced peptide bond pseudopeptide Michael acceptors for the treatment of Human African Trypanosomiasis. Molecules 2022, 27, 3765. [Google Scholar] [CrossRef] [PubMed]






| Cmps | 0.25 × IC50 | 0.5 × IC50 | IC50 | 2 × IC50 | 4 × IC50 |
|---|---|---|---|---|---|
| CD24 | 0.05 µM | 0.1 µM | 0.2 µM | 0.4 µM | 0.8 µM |
| Curcumin | 3.07 µM | 6.15 µM | 12.3 µM | 24.6 µM | 49.2 µM |
| CD24 + Curcumin | 0.05 ± 3.07 µM | 0.1 ± 6.15 µM | 0.2 ± 12.3 µM | 0.4 ± 24.6 µM | 0.8 ± 49.2 µM |
| Fraction Affected (fa) | % of Rhodesain Inhibition | Combination Index (CI) | Diagnosis of Combined Effect |
|---|---|---|---|
| 0.50 | 50% | 1.08 | Additive |
| 0.60 | 60% | 0.93 | Synergism |
| 0.70 | 70% | 0.81 | Synergism |
| 0.80 | 80% | 0.70 | Synergism |
| 0.90 | 90% | 0.59 | Synergism |
| 1 | 100% | 0.45 | Synergism |
| Compounds | T. b. brucei EC50 µM | HEK293 EC50 µM | SI |
|---|---|---|---|
| CD24 [29] | 10.1 ± 0.5 | >70 | >6.9 |
| Curcumin [31] | 3.12 ± 0.43 | >70 | >22.4 |
| CD24 + curcumin | 4.85 ± 0.02 | >70 | >14.4 |
| Fexinidazole [38] | 2.38 ± 0.88 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Chio, C.; Previti, S.; De Luca, F.; Bogacz, M.; Zimmer, C.; Wagner, A.; Schirmeister, T.; Zappalà, M.; Ettari, R. Drug Combination Studies of the Dipeptide Nitrile CD24 with Curcumin: A New Strategy to Synergistically Inhibit Rhodesain of Trypanosoma brucei rhodesiense. Int. J. Mol. Sci. 2022, 23, 14470. https://doi.org/10.3390/ijms232214470
Di Chio C, Previti S, De Luca F, Bogacz M, Zimmer C, Wagner A, Schirmeister T, Zappalà M, Ettari R. Drug Combination Studies of the Dipeptide Nitrile CD24 with Curcumin: A New Strategy to Synergistically Inhibit Rhodesain of Trypanosoma brucei rhodesiense. International Journal of Molecular Sciences. 2022; 23(22):14470. https://doi.org/10.3390/ijms232214470
Chicago/Turabian StyleDi Chio, Carla, Santo Previti, Fabiola De Luca, Marta Bogacz, Collin Zimmer, Annika Wagner, Tanja Schirmeister, Maria Zappalà, and Roberta Ettari. 2022. "Drug Combination Studies of the Dipeptide Nitrile CD24 with Curcumin: A New Strategy to Synergistically Inhibit Rhodesain of Trypanosoma brucei rhodesiense" International Journal of Molecular Sciences 23, no. 22: 14470. https://doi.org/10.3390/ijms232214470
APA StyleDi Chio, C., Previti, S., De Luca, F., Bogacz, M., Zimmer, C., Wagner, A., Schirmeister, T., Zappalà, M., & Ettari, R. (2022). Drug Combination Studies of the Dipeptide Nitrile CD24 with Curcumin: A New Strategy to Synergistically Inhibit Rhodesain of Trypanosoma brucei rhodesiense. International Journal of Molecular Sciences, 23(22), 14470. https://doi.org/10.3390/ijms232214470


_Putnam.png)