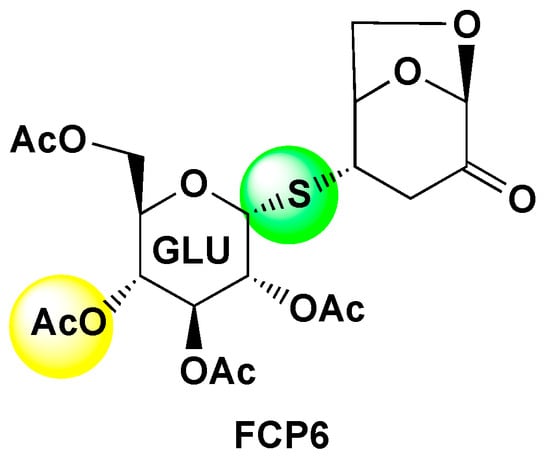
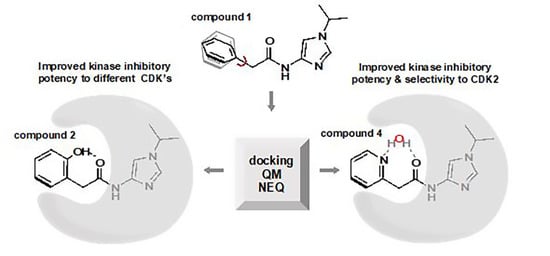
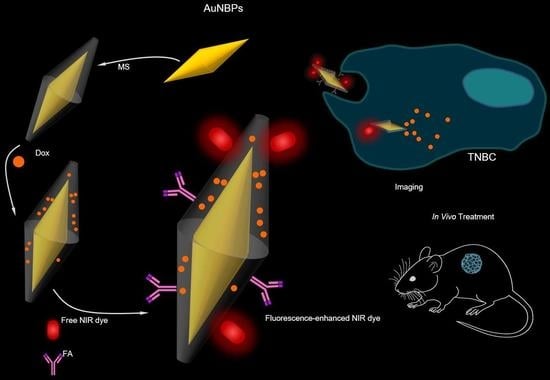
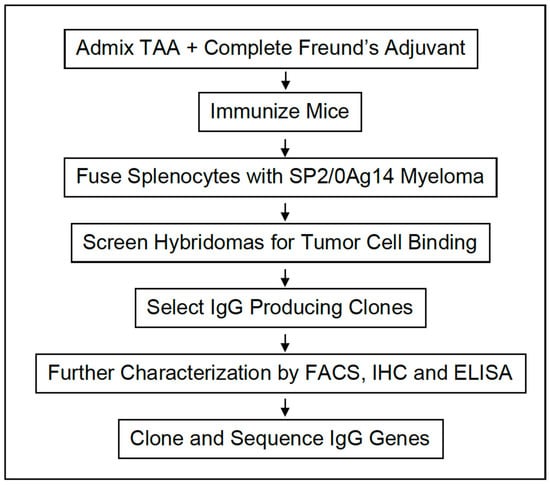
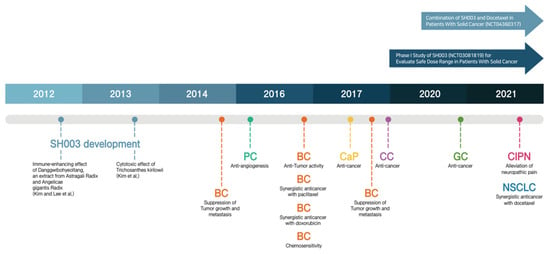
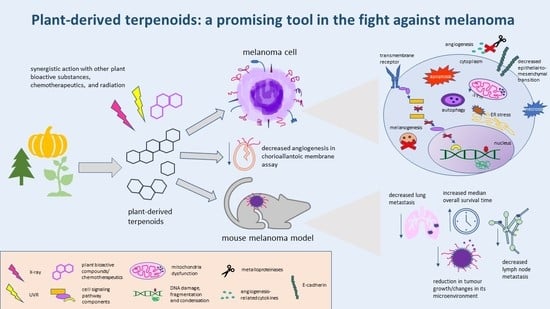
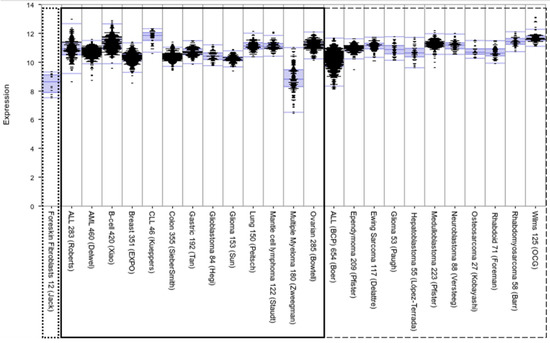
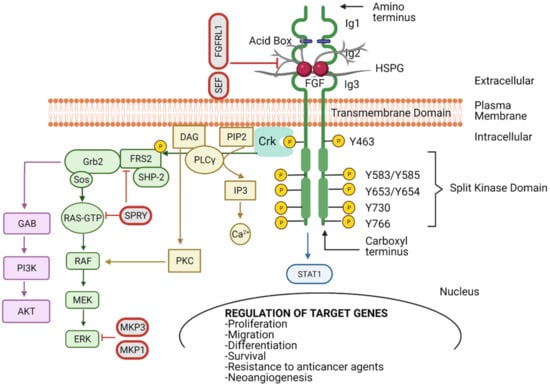
The Development of Anti-cancer Agents
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Cancer Drug Development".
Viewed by 56941Editor
Interests: therapeutic and diagnostic antibodies; cancer therapy; discovery of therapeutic targets
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
In the past few decades, advances in unraveling the complex biology of human cancer, the immune system, and the tumor microenvironment have been translated into the development of patient-tailored therapeutic agents, which ultimately cure and or improved survival for many patients with various types of cancer. In contrast, for other cancers, there is still either no effective treatment (e.g., pancreatic cancer) or the response can be of short duration. One of the major contributors to this poor response (i.e., primary resistance or acquired resistance) to anti-cancer drugs is the heterogeneous nature of human cancers. It is essential to discover additional therapeutic targets and to develop more effective and less toxic therapeutic agents for patients diagnosed at different stages of cancer. This is an active area of research.
This Special Issue will highlight some key advances in the development of various types of therapeutic agents in cancer from monoclonal antibody-based drugs, to small-molecule tyrosine kinase inhibitors, drugs-targeting hormone-dependent tumors, cell-cycle inhibitors, and the CAR-T cells. The outstanding challenges and future opportunities for the discovery of novel therapeutic targets and the development of more effective therapeutic agents will also be highlighted.
Prof. Helmout Modjtahedi
Guest Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- monoclonal antibody-based drugs
- small-molecule tyrosine kinase inhibitors
- drugs-targeting hormone-dependent tumors
- cell-cycle inhibitors
- CAR-T cells
- cancers