TNF-α Blocker Effect of Naringenin-Loaded Sericin Microparticles that Are Potentially Useful in the Treatment of Psoriasis
Abstract
:1. Introduction

2. Results
2.1. Preparation, Characterization and Analysis of Naringenin-Loaded Sericin Microspheres

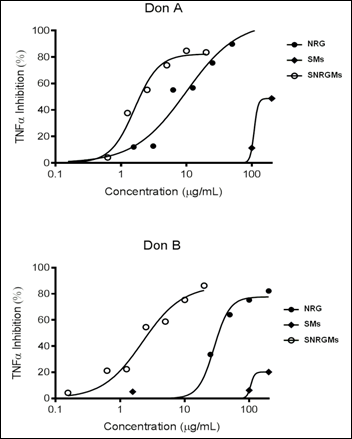
2.2. Biological Evaluation

| Concentration (µg/mL) | Percent Inhibition of Cell Viability (AFU Fluorescence ± SD) | |||||
|---|---|---|---|---|---|---|
| Donor A | Donor B | |||||
| SMs | NRG | SNRGMs | SMs | NRG | SNRGMs | |
| 0.0 | (216,176 ± 16,662) | (216,176 ± 16,662) | (216,176 ± 16,662) | (296,090 ± 3485) | (296,090 ± 3485) | (296,090 ± 3485) |
| 1.5 | −0.6 (217,369 ± 23,816) | 12.7 (188,915 ± 5021) | 12.0 (190,284 ± 17,929) | 5.7 (279,202 ± 19,030) | 7.3 (274,536 ± 15,953) | 2.8 (287,884 ± 9992) |
| 3.1 | 3.4 (208,859 ± 12,809) | 7.7 (199,427 ± 12,120) | 13.5 (186,912 ± 19,949) | −0.5 (297,628 ± 2021) | 3.9 (284,657 ± 10,232) | 3.8 (284,955 ± 15,984) |
| 6.2 | −1.7 (219,873 ± 3647) | 4.9 (205,540 ± 2716) | 9.1 (196,516 ± 7684) | −0.2 (296,641 ± 8079) | 4.0 (284,141 ± 8173) | −1.3 (299,915 ± 5812) |
| 12.5 | 6.6 (201,875 ± 23,690) | −7.4 (232,158 ± 8719) | −3.6 (224,022 ± 8827) | 12.0 (260,699 ± 21,230) * | 2.4 (288,952 ± 12,039) | −5.6 (312,652 ± 817) |
| 25 | 14.4 (185,018 ± 8764) | −8.1 (233,665 ± 14,492) | −17.5 (254,054 ± 5993) * | 8.4 (271,324 ± 22,478) | −2.1 (302,298 ± 5691) | −6.8 (316,310 ± 15,836) |
| 50 | −0.4 (216,983 ± 18,889) | 0.4 (215,207 ± 10,787) | −11.0 (239,937 ± 17,440) | 4.0 (284,183 ± 20,694) | −4.5 (309,485 ± 6492) | −10.2 (326,391 ± 9586) |
| 100 | −2.9 (222,453 ± 17,007) | 9.1 (196,415 ± 9256) | −18.6 (256,290 ± 6843) | −2.4 (303,159 ± 29,542) | 4.1 (284,065 ± 10,582) | −12.1 (331,907 ± 6113) * |
| 200 | −20.6 (260,790 ± 23,226) ** | 26.6 (158,719 ± 12,585) *** | 73.1 (58,242 ± 7469) *** | −3.3 (305,767 ± 23,492) | 23.7 (225,848 ± 12,064) *** | 67.2 (97,067 ± 27,795) *** |
| Treatment | IC50 (μg/mL) | |
|---|---|---|
| Donor A | Donor B | |
| SMs | 200 * | >200 ** |
| NRG | 8.7 | 33.6 |
| SNRGMs *** | 1.9 | 2.8 |
| Fold increase | 4.6 | 12.0 |
3. Discussion
4. Experimental Section
4.1. Materials

4.2. Preparation of Sericin and Naringenin-Loaded Sericin Microspheres
4.3. Granulometric Analysis
4.4. HPLC Analysis of NRG-Loaded Sericin Microspheres
4.5. Encapsulation Efficiency Determination
4.6. Human Peripheral Blood Mononuclear Cells Preparation
4.7. Cytotoxicity Assay

4.8. TNF-α Inhibition Assay
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gaggeri, R.; Rossi, D.; Hajikarimian, N.; Martino, E.; Bracco, F.; Grisoli, P.; Dacarro, C.; Leoni, F.; Mascheroni, G.; Collina, S.; et al. Preliminary study on TNFα-blocker activity of amygdalus lycioides spach extracts. Open Nat. Prod. J. 2010, 3, 20–25. [Google Scholar] [CrossRef]
- Gaggeri, R.; Rossi, D.; Christodoulou, M.S.; Passerella, D.; Leoni, F.; Azzolina, O.; Collina, S. Chiral flavanones from amygdalus lycioides spach: Structural elucidation and identification of TNFα inhibitors by bioactivity-guided fractionation. Molecules 2012, 17, 1665–1674. [Google Scholar] [CrossRef]
- Gaggeri, R.; Rossi, D.; Daglia, M.; Leoni, F.; Avanzini, M.A.; Mantelli, M.; Juza, M.; Collina, S. An ecofriendly enantioselective access to (R)-Naringenin as inhibitor of pro-inflammatory cytokines release. Chem. Biodivers. 2013, 10, 1532–1538. [Google Scholar]
- Padamwar, M.N.; Pawar, A.P. Silk sericin and its applications: A review. J. Sci. Ind. Res. 2004, 63, 323–329. [Google Scholar]
- Chlapanidas, T.; Faragò, S.; Lucconi, G.; Perteghella, S.; Galuzzi, M.; Mantelli, M.; Avanzini, M.A.; Tosca, M.C.; Marazzi, M.; Vigo, D.; et al. Sericins exhibit ROS-scavenging, anti-tyrosinase, anti-elastase, and in vitro immunomodulatory activities. Int. J. Biol. Macromol. 2013, 58, 47–56. [Google Scholar] [CrossRef]
- Prieto-Pérez, P.; Cabaleiro, T.; Daudén, E.; Ochoa, M.; Roman, M.; Abad-Santos, F. Genetics of psoriasis and pharmacogenetics of biological drugs. Autoimmun. Dis. 2013, 2013, 613086. [Google Scholar]
- Traub, M.; Marshall, K. Psoriasis—Pathophysiology, conventional, and alternative approaches to treatment. Altern. Med. Rev. 2007, 12, 319–330. [Google Scholar]
- Nograles, K.E.; Krueger, J.G. Anti-cytokine therapies for psoriasis. Exp. Cell Res. 2011, 317, 1293–1300. [Google Scholar] [CrossRef]
- Bos, J.D.; Spuls, P.I. Topical treatments in psoriasis: Today and tomorrow. Clin. Dermatol. 2008, 26, 432–437. [Google Scholar] [CrossRef]
- Laws, P.M.; Young, H.S. Current and emerging systemic treatment strategies for psoriasis. Drugs 2012, 72, 1867–1880. [Google Scholar] [CrossRef]
- Kupetsky, E.A.; Mathers, A.R.; Ferris, L.K. Anti-cytokine therapy in the treatment of psoriasis. Cytokine 2013, 61, 704–712. [Google Scholar] [CrossRef]
- Oh, H.; Kim, M.K.; Lee, K.H. Preparation of sericin microparticles by electrohydrodynamic spraying and their application in drug delivery. Macromol. Res. 2011, 19, 266–272. [Google Scholar] [CrossRef]
- Leoni, F.; Fossati, G.; Lewis, E.C.; Lee, J.K.; Porro, G.; Pagani, P.; Modena, D.; Moras, M.L.; Pozzi, P.; Reznikov, L.L.; et al. The histone deacetylase inhibitor ITF2357 reduces production of pro-inflammatory cytokines in vitro and systemic inflammation in vivo. Mol. Med. 2005, 11, 1–15. [Google Scholar]
- Padamwar, M.N.; Pawar, A.P.; Daithankar, A.V.; Mahadik, K.R. Silk sericin as a moisturizer: An in vivo study. J. Cosmet. Dermatol. 2005, 4, 250–257. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chlapanidas, T.; Perteghella, S.; Leoni, F.; Faragò, S.; Marazzi, M.; Rossi, D.; Martino, E.; Gaggeri, R.; Collina, S. TNF-α Blocker Effect of Naringenin-Loaded Sericin Microparticles that Are Potentially Useful in the Treatment of Psoriasis. Int. J. Mol. Sci. 2014, 15, 13624-13636. https://doi.org/10.3390/ijms150813624
Chlapanidas T, Perteghella S, Leoni F, Faragò S, Marazzi M, Rossi D, Martino E, Gaggeri R, Collina S. TNF-α Blocker Effect of Naringenin-Loaded Sericin Microparticles that Are Potentially Useful in the Treatment of Psoriasis. International Journal of Molecular Sciences. 2014; 15(8):13624-13636. https://doi.org/10.3390/ijms150813624
Chicago/Turabian StyleChlapanidas, Theodora, Sara Perteghella, Flavio Leoni, Silvio Faragò, Mario Marazzi, Daniela Rossi, Emanuela Martino, Raffaella Gaggeri, and Simona Collina. 2014. "TNF-α Blocker Effect of Naringenin-Loaded Sericin Microparticles that Are Potentially Useful in the Treatment of Psoriasis" International Journal of Molecular Sciences 15, no. 8: 13624-13636. https://doi.org/10.3390/ijms150813624
APA StyleChlapanidas, T., Perteghella, S., Leoni, F., Faragò, S., Marazzi, M., Rossi, D., Martino, E., Gaggeri, R., & Collina, S. (2014). TNF-α Blocker Effect of Naringenin-Loaded Sericin Microparticles that Are Potentially Useful in the Treatment of Psoriasis. International Journal of Molecular Sciences, 15(8), 13624-13636. https://doi.org/10.3390/ijms150813624