Long-Term Brain Disorders in Post Covid-19 Neurological Syndrome (PCNS) Patient
Abstract
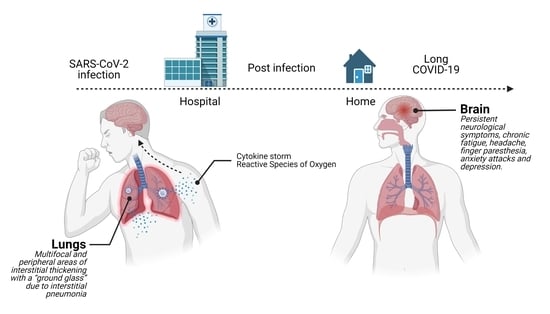
:1. Introduction
2. Case
2.1. Hospital Phase
2.2. Post Hospital Phase
3. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef] [Green Version]
- Carfì, A.; Bernabei, R.; Landi, F. Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Rudroff, T.; Fietsam, A.C.; Deters, J.R.; Bryant, A.D.; Kamholz, J. Post-COVID-19 Fatigue: Potential Contributing Factors. Brain Sci. 2020, 10, 1012. [Google Scholar] [CrossRef] [PubMed]
- Morgul, E.; Bener, A.; Atak, M.; Akyel, S.; Aktas, S.; Bhugra, D.; Ventriglio, A.; Jordan, T.R. COVID-19 pandemic and psy-chological fatigue in Turkey. Int. J. Soc. Psychiatry 2020, 1–8. [Google Scholar] [CrossRef]
- Satici, B.; Gocet-Tekin, E.; Deniz, M.E.; Satici, S.A. Adaptation of the Fear of COVID-19 Scale: Its Association with Psychological Distress and Life Satisfaction in Turkey. Int. J. Ment. Health Addict. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C.; Anrather, J. The immunology of stroke: From mechanisms to translation. Nat. Med. 2011, 17, 796–808. [Google Scholar] [CrossRef] [PubMed]
- Anrather, J.; Iadecola, C. Inflammation and Stroke: An Overview. Neurotherapeutics 2016, 13, 661–670. [Google Scholar] [CrossRef]
- Iadecola, C.; Anrather, J.; Kamel, H. Effects of COVID-19 on the Nervous System. Cell 2020, 183, 16–27.e1. [Google Scholar] [CrossRef]
- Nuzzo, D.; Picone, P. Potential neurological effects of severe COVID-19 infection. Neurosci. Res. 2020, 158, 1–5. [Google Scholar] [CrossRef]
- Needham, E.; Newcombe, V.; Michell, A.; Thornton, R.; Grainger, A.; Anwar, F.; Warburton, E.; Menon, D.; Trivedi, M.; Sawcer, S. Mononeuritis multiplex: An unexpectedly frequent feature of severe COVID-19. J. Neurol. 2020, 1–5. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, Y.; Liang, X.; Gao, B.; Liu, M.; Li, W.; Chen, Z.; Wang, Z. COVID-19 Associated Ischemic Stroke and Hemorrhagic Stroke: Incidence, Potential Pathological Mechanism, and Management. Front. Neurol. 2020, 11, 571996. [Google Scholar] [CrossRef] [PubMed]
- Zakeri, A.; Jadhav, A.P.; Sullenger, B.A.; Nimjee, S.M. Ischemic stroke in COVID-19-positive patients: An overview of SARS-CoV-2 and thrombotic mechanisms for the neurointerventionalist. Neurointerv. Surg. 2021, 13, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.K.; Paliwal, V.K.; Gupta, A. Encephalopathy in patients with COVID-19: A review. J. Med. Virol. 2021, 93, 206–222. [Google Scholar] [CrossRef]
- Wijeratne, T.; Crewther, S. Post-COVID 19 Neurological Syndrome (PCNS); a novel syndrome with challenges for the global neurology community. J. Neurol. Sci. 2020, 419, 117179. [Google Scholar] [CrossRef]
- Wijeratne, T.; Crewther, S. COVID-19 and long-term neurological problems: Challenges ahead with Post-COVID-19 Neuro-logical Syndrome. Aust. J. Gen. Pract. 2021. [Google Scholar] [CrossRef]
- Wijeratne, T.; Wijeratne, C. Clinical utility of serial systemic immune inflammation indices (SSIIi) in the context of post covid-19 neurological syndrome (PCNS). J. Neurol. Sci. 2021, 423, 117356. [Google Scholar] [CrossRef]
- Neumann, B.; Schmidbauer, M.L.; Dimitriadis, K.; Otto, S.; Knier, B.; Niesen, W.-D.; Hosp, J.A.; Günther, A.; Lindemann, S.; Nagy, G.; et al. Cerebrospinal fluid findings in COVID-19 patients with neurological symptoms. J. Neurol. Sci. 2020, 418, 117090. [Google Scholar] [CrossRef]
- Nuzzo, D.; Vasto, S.; Scalisi, L.; Cottone, S.; Cambula, G.; Manfredi, R.; Giacomazza, D.; Picone, P. Post-acute COVID-19 neurological syndrome: A new medical challenge. J. Clin. Med. under review.
- Cantuti-Castelvetri, L.; Ojha, R.; Pedro, L.D.; Djannatian, M.; Franz, J. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science 2020, 13, 856–860. [Google Scholar] [CrossRef]
- Liotta, E.M.; Batra, A.; Clark, J.R.; Shlobin, N.A.; Hoffman, S.C.; Orban, Z.S.; Koralnik, I.J. Frequent neurologic manifestations and en-cephalopathy-associated morbidity in Covid-19 patients. Ann. Clin. Transl. Neurol. 2020, 7, 2221–2230. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.I.; Abd-Allah, F.; Al-Senani, F.; Aytac, E.; Borhani-Haghighi, A.; Ciccone, A.; Gomez, C.R.; Gurkas, E.; Hsu, C.Y.; Jani, V.; et al. Management of acute ischemic stroke in patients with COVID-19 infection: Report of an international panel. Int. J. Stroke 2020, 15, 540–554. [Google Scholar] [CrossRef] [PubMed]
- Webb, S.; Wallace, V.C.; Martin-Lopez, D.; Yogarajah, M. Guillain-Barré syndrome following COVID-19: A newly emerging post-infectious complication. BMJ Case Rep. 2020, 13, e236182. [Google Scholar] [CrossRef] [PubMed]
- Harapan, B.N.; Yoo, H.J. Neurological symptoms, manifestations, and complications associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease 19 (COVID-19). J. Neurol. 2021. [Google Scholar] [CrossRef]




| Characteristics | Pre Infection December | After Intensive Care April | Post Infection September |
|---|---|---|---|
| Weight (Kg) | 104 | 75.8 | 81.1 |
| BMI | 34 | 25.7 | 28.1 |
| Fat (%) | 37.4 | 24.8 | 30.4 |
| Status | Obese | Normal weight | Overweight |
| Diseases | No | … | Neurological disorders |
| Psychiatric activity | Normal | … | Depression |
| 6-MWT (meter) | 620 | <1 | 510 |
| Chest (cm) | 128 | 105 | 107 |
| Waist (cm) | 141 | 103 | 104 |
| Biceps (cm) | 31 | 25 | 27 |
| Leg (cm) | 51 | 45 | 47 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nuzzo, D.; Cambula, G.; Bacile, I.; Rizzo, M.; Galia, M.; Mangiapane, P.; Picone, P.; Giacomazza, D.; Scalisi, L. Long-Term Brain Disorders in Post Covid-19 Neurological Syndrome (PCNS) Patient. Brain Sci. 2021, 11, 454. https://doi.org/10.3390/brainsci11040454
Nuzzo D, Cambula G, Bacile I, Rizzo M, Galia M, Mangiapane P, Picone P, Giacomazza D, Scalisi L. Long-Term Brain Disorders in Post Covid-19 Neurological Syndrome (PCNS) Patient. Brain Sciences. 2021; 11(4):454. https://doi.org/10.3390/brainsci11040454
Chicago/Turabian StyleNuzzo, Domenico, Gaetano Cambula, Ignazio Bacile, Manfredi Rizzo, Massimo Galia, Paola Mangiapane, Pasquale Picone, Daniela Giacomazza, and Luca Scalisi. 2021. "Long-Term Brain Disorders in Post Covid-19 Neurological Syndrome (PCNS) Patient" Brain Sciences 11, no. 4: 454. https://doi.org/10.3390/brainsci11040454
APA StyleNuzzo, D., Cambula, G., Bacile, I., Rizzo, M., Galia, M., Mangiapane, P., Picone, P., Giacomazza, D., & Scalisi, L. (2021). Long-Term Brain Disorders in Post Covid-19 Neurological Syndrome (PCNS) Patient. Brain Sciences, 11(4), 454. https://doi.org/10.3390/brainsci11040454