A Study on the Structure and Biomedical Application Characteristics of Phosphate Coatings on ZKX500 Magnesium Alloys
Abstract
:1. Introduction
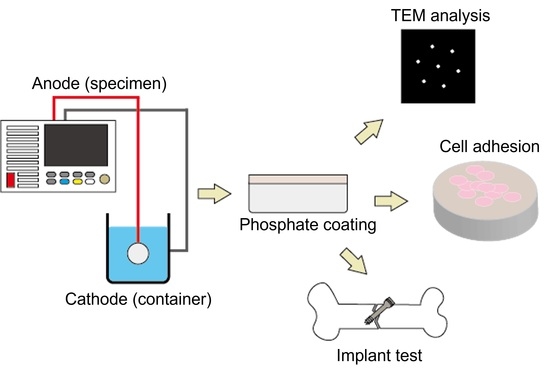
2. Materials and Methods
2.1. Materials and Specimen Preparation
2.2. Characterization of the Coatings
2.3. In Vitro Degradation Test and Mechanical Properties
2.4. Biocompatibility Analysis
2.5. Implantation Test
3. Results and Discussion
3.1. Micromorphology and Composition of the Coating
3.2. Degradation Rates and Models
3.3. Mechanical Properties after Immersion
3.4. Cell Adhesion
3.5. Animal Experiment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, J.L.; Xu, J.K.; Hopkins, C.; Chow, D.H.K.; Qin, L. Biodegradable magnesium-based implants in orthopedics—A general review and perspectives. Adv. Sci. 2020, 7, 1902443. [Google Scholar] [CrossRef] [Green Version]
- Jamel, M.M.; Jamel, M.M.; Lopez, H.F. Designing advanced biomedical biodegradable Mg alloys: A review. Metals 2022, 12, 85. [Google Scholar] [CrossRef]
- Sezer, N.; Evis, Z.; Kayhan, S.M.; Tahmasebifar, A.; Koç, M. Review of magnesium-based biomaterials and their applications. J. Magnes. Alloy. 2018, 6, 23–43. [Google Scholar] [CrossRef]
- Lee, J.W.; Han, H.S.; Han, K.J.; Park, J.; Jeon, H.; Ok, M.R.; Seok, H.K.; Ahn, J.P.; Lee, K.E.; Lee, D.H.; et al. Long-term clinical study and multiscale analysis of in vivo biodegradation mechanism of Mg alloy. Proc. Natl. Acad. Sci. USA 2016, 113, 716–721. [Google Scholar] [CrossRef] [Green Version]
- Tan, L.; Yu, X.; Wan, P.; Yang, K. Biodegradable materials for bone repairs: A review. J. Mater. Sci. Technol. 2013, 29, 503–513. [Google Scholar] [CrossRef]
- Jin, W.; Wang, G.; Lin, Z.; Feng, H.; Li, W.; Peng, X.; Qasim, A.M.; Chu, P.K. Corrosion resistance and cytocompatibility of tantalum-surface-functionalized biomedical ZK60 Mg alloy. Corros. Sci. 2017, 114, 45–56. [Google Scholar] [CrossRef]
- Song, G. Control of biodegradation of biocompatable magnesium alloys. Corros. Sci. 2007, 49, 1696–1701. [Google Scholar] [CrossRef]
- Ma, C.; Liu, M.; Wu, G.; Ding, W.; Zhu, Y. Tensile properties of extruded ZK60-RE alloys. Mater. Sci. Eng. A 2003, 349, 207–212. [Google Scholar] [CrossRef]
- Uddin, M.S.; Hall, C.; Murphy, P. Surface treatments for controlling corrosion rate of biodegradable Mg and Mg-based alloy implants. Sci. Technol. Adv. Mater. 2015, 16, 24. [Google Scholar] [CrossRef] [Green Version]
- Cai, S.; Lei, T.; Li, N.; Feng, F. Effects of Zn on microstructure, mechanical properties and corrosion behavior of Mg-Zn alloys. Mater. Sci. Eng. C 2012, 32, 2570–2577. [Google Scholar] [CrossRef]
- Sun, M.; Wu, G.; Wang, W.; Ding, W. Effect of Zr on the microstructure, mechanical properties and corrosion resistance of Mg-10Gd-3Y magnesium alloy. Mater. Sci. Eng. A 2009, 523, 145–151. [Google Scholar] [CrossRef]
- Yin, P.; Li, N.F.; Lei, T.; Liu, L.; Ouyang, C. Effects of Ca on microstructure, mechanical and corrosion properties and biocompatibility of Mg-Zn-Ca alloys. J. Mater. Sci. Mater. Med. 2013, 24, 1365–1373. [Google Scholar] [CrossRef]
- Chen, Y.T.; Hung, F.Y.; Lin, Y.L.; Lin, C.Y. Biodegradation ZK50 Magnesium Alloy Compression Screws: Mechanical Properties, Biodegradable Characteristics and Implant Test. J. Orthop. Sci. 2020, 25, 1107–1115. [Google Scholar] [CrossRef]
- Huang, Y.-T.; Hung, F.-Y.; Kuan, F.-C.; Hsu, K.-L.; Su, W.-R.; Lin, C.-Y. Microstructure, Mechanical Properties, Degradation Behavior, and Implant Testing of Hot-Rolled Biodegradable ZKX500 Magnesium Alloy. Appl. Sci. 2021, 11, 10677. [Google Scholar] [CrossRef]
- Chaharmahali, R.; Fattah-Alhosseini, A.; Babaei, K. Surface characterization and corrosion behavior of calcium phosphate (Ca-P) base composite layer on Mg and its alloys using plasma electrolytic oxidation (PEO): A review. J. Magnes. Alloy. 2021, 9, 21–40. [Google Scholar] [CrossRef]
- Horynová, M.; Remešová, M.; Klakurková, L.; Dvořák, K.; Ročňáková, I.; Yan, S.; Čelko, L.; Song, G.-L. Design of tailored biodegradable implants: The effect of voltage on electrodeposited calcium phosphate coatings on pure magnesium. J. Am. Ceram. Soc. 2019, 102, 123–135. [Google Scholar] [CrossRef] [Green Version]
- Prakash, C.; Singh, S.; Pabla, B.; Uddin, M. Synthesis, characterization, corrosion and bioactivity investigation of nano-HA coating deposited on biodegradable Mg-Zn-Mn alloy. Surf. Coat. Technol. 2018, 346, 9–18. [Google Scholar] [CrossRef]
- ASTM G31-04(2004); Standard Practice for Laboratory Immersion Corrosion Testing of Metals. ASTM International: West Conshohocken, PA, USA, 2004.
- ISO 10993-5:2009; Biological Evaluation of Medical Devices-Part 5: Tests for in vitro Cytotoxicity. International Organization for Standardization (ISO): Geneva, Switzerland, 2009.
- Rozaini, M.Z.H.; Hamzah, H.; Wai, C.P.; Razali, M.H.; Osman, U.M.; Anuar, S.T.; Soh, S.K.C.; Ghazali, S.R.B.; Ibrahim, N.H.; Fei, L.C.; et al. Calcium Hydroxyapatite-based Marine Origin: Novel Sunscreen Materials for Cosmeceutical Treatments. Orient. J. Chem. 2018, 34, 2770–2776. [Google Scholar] [CrossRef] [Green Version]
- Oyane, A.; Kim, H.M.; Furuya, T.; Kokubo, T.; Miyazaki, T.; Nakamura, T. Preparation and assessment of revised simulated body fluids. J. Biomed. Mater. Res. Part A 2003, 65, 188–195. [Google Scholar] [CrossRef]
- Vinogradov, A.; Orlov, D.; Estrin, Y. Improvement of fatigue strength of a Mg-Zn-Zr alloy by integrated extrusion and equal- channel angular pressing. Scr. Mater. 2012, 67, 209–212. [Google Scholar] [CrossRef]
- Callister, D.W.; Rethwisch, D.G. Materials Science and Engineering: An Introduction; Wiley: New York, NY, USA, 2018; Volume 9. [Google Scholar]
- Huang, Z.H.; Qi, W.J.; Jing, X.U. Effect of microstructure on impact toughness of magnesium alloys. Trans. Nonferr. Met. Soc. China 2012, 22, 2334–2342. [Google Scholar] [CrossRef]
- Liao, J.; Hotta, M.; Kaneko, K.; Kondoh, K. Enhanced impact toughness of magnesium alloy by grain refinement. Scr. Mater. 2009, 61, 208–211. [Google Scholar] [CrossRef]
- Eliezer, A.; Gutman, E.M.; Abramov, E.; Unigovski, Y. Corrosion fatigue of die-cast and extruded magnesium alloys. J. Light Met. 2001, 1, 179–186. [Google Scholar] [CrossRef]
- He, C.; Liu, Y.; Li, J.; Yang, K.; Wang, Q.; Chen, Q. Very-high-cycle fatigue crack initiation and propagation behaviours of magnesium alloy ZK60. Mater. Sci. Technol. 2018, 34, 639–647. [Google Scholar] [CrossRef]
- Wang, Z.X.; Xu, L.; Zhang, J.W.; Ye, F.; Lv, W.G.; Xu, C.; Lu, S.; Yang, J. Preparation and Degradation Behavior of Composite Bio-Coating on ZK60 Magnesium Alloy Using Combined Micro-Arc Oxidation and Electrophoresis Deposition. Front. Mater. 2020, 7, 14. [Google Scholar] [CrossRef]
- Rahim, S.A.; Nikhil, T.T.; Joseph, M.A.; Hanas, T. In vitro degradation and mechanical behaviour of calcium phosphate coated Mg-Ca alloy. Mater. Technol. 2021, 36, 738–746. [Google Scholar] [CrossRef]
- Yu, J.I. Mg Alloy Surface Treatment. In Magnesium Alloys; Aliofkhazraei, M., Ed.; Intech Europe: Rijeka, Croatia, 2017; pp. 75–90. [Google Scholar]
- Lee, J.L.; Jian, S.Y.; Kuo, K.N.; You, J.L.; Lai, Y.T. Effect of Surface Properties on Corrosion Resistance of ZK60 Mg Alloy Microarc Oxidation Coating. IEEE Trans. Plasma Sci. 2019, 47, 1172–1180. [Google Scholar] [CrossRef]
- Li, B.; Han, Y.; Qi, K. Formation mechanism, degradation behavior, and cytocompatibility of a nanorod-shaped HA and pore-sealed MgO bilayer coating on magnesium. ACS Appl. Mater. Interfaces 2014, 6, 18258–18274. [Google Scholar] [CrossRef]
- Jamesh, M.I.; Wu, G.; Zhao, Y.; McKenzie, D.R.; Bilek, M.M.; Chu, P.K. Effects of zirconium and oxygen plasma ion implantation on the corrosion behavior of ZK60 Mg alloy in simulated body fluids. Corros. Sci. 2014, 82, 7–26. [Google Scholar] [CrossRef]
- Chen, X.; Birbilis, N.; Abbott, T. Review of corrosion-resistant conversion coatings for magnesium and its alloys. Corrosion 2011, 67, 035005-1–035005-16. [Google Scholar] [CrossRef]
- Li, K.; Wang, B.; Yan, B.; Lu, W. Microstructure, invitro corrosion and cytotoxicity of Ca-P coatings on ZK60 magnesium alloy prepared by simple chemical conversion and heat treatment. J. Biomater. Appl. 2013, 28, 375–384. [Google Scholar] [CrossRef]



























| Compound | Amount (g/L) |
|---|---|
| NaCl | 5.403 |
| NaHCO3 | 0.740 |
| Na2CO3 | 2.046 |
| KCl | 0.225 |
| K2HPO4 | 0.230 |
| MgCl2·6H2O | 0.311 |
| HEPES | 11.928 |
| CaCl2 | 0.294 |
| Na2SO4 | 0.072 |
| NaOH | Buffer to pH 7.4 |
| Months | Mg Screw Length (mm) |
|---|---|
| 1 | 12.25 |
| 2 | 9.30 |
| 3 | 9.18 |
| 4 | 8.98 |
| 5 | 8.01 |
| 6 | 7.77 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-T.; Wu, W.-Y.; Hung, F.-Y.; Kuan, F.-C.; Hsu, K.-L.; Su, W.-R.; Yen, C.-W. A Study on the Structure and Biomedical Application Characteristics of Phosphate Coatings on ZKX500 Magnesium Alloys. Bioengineering 2022, 9, 542. https://doi.org/10.3390/bioengineering9100542
Huang Y-T, Wu W-Y, Hung F-Y, Kuan F-C, Hsu K-L, Su W-R, Yen C-W. A Study on the Structure and Biomedical Application Characteristics of Phosphate Coatings on ZKX500 Magnesium Alloys. Bioengineering. 2022; 9(10):542. https://doi.org/10.3390/bioengineering9100542
Chicago/Turabian StyleHuang, Ying-Ting, Wen-Yu Wu, Fei-Yi Hung, Fa-Chuan Kuan, Kai-Lan Hsu, Wei-Ren Su, and Chen-Wei Yen. 2022. "A Study on the Structure and Biomedical Application Characteristics of Phosphate Coatings on ZKX500 Magnesium Alloys" Bioengineering 9, no. 10: 542. https://doi.org/10.3390/bioengineering9100542
APA StyleHuang, Y. -T., Wu, W. -Y., Hung, F. -Y., Kuan, F. -C., Hsu, K. -L., Su, W. -R., & Yen, C. -W. (2022). A Study on the Structure and Biomedical Application Characteristics of Phosphate Coatings on ZKX500 Magnesium Alloys. Bioengineering, 9(10), 542. https://doi.org/10.3390/bioengineering9100542