Anti-carcinogenic Effects of the Flavonoid Luteolin
Abstract
:1. Introduction
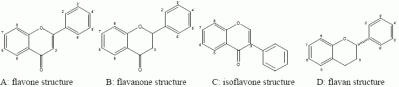
2. Flavonoids: some functionally relevant aspects of molecular structure

3. Pharmacological Effects
| Target | Effect of luteolin | Publication source(s) |
|---|---|---|
| Tumor cell proliferation | Inhibits proliferation of various tumor cell lines in vitro | [12,13,14,15,16,17,18,19,20,21,22,23,25,27,28,30,31,32,34,35] |
| Cell cycle arrest | Induces cell cycle arrest in G2/M, S, or G0/1 phase | [19,22,23,24,25,26] |
| Angiogenesis | Inhibits bFGF and VEGF induced in vitro angiogenesis of BBE cells | [12] |
| Tumor growth in vivo | Inhibits growth of tumors from different organs when applied p.o. or i.p. | [21,33,34,35,36,37,38,40,50,68] |
| Anti-oxidant enzymes and compounds | Increases levels of SOD, CAT, GPx, Vit A, Vit C, ß-Carotene | [29,49,50,51] |
| NAT | Inhibits N-acetyltransferase in cancer cell lines | [54,56,57] |
| MMP | Inhibits secretion of MMP-2 and MMP-9 release | [20] |
| IGF / IFR | Inhibits tyrosin phosphorylation of IGFR | [36] |
| EGF / EGFR | Inhibits EGF or IGF-1 induced activation of EGFR | [20,36,66] |
| HGF / c-Met | Inhibits phosphorylation of hepatocyte growth factor receptor c-Met | [58] |
| AR | Downregulates androgen receptor | [35] |
| PI-3-Kinase / Akt | Inhibits Akt phosphorylation | [36,65,66,67] |
| PI-3-Kinase / Akt | Suppresses Akt expression | [21] |
| Mitochondria | Reduces mitochondrial membrane potential | [21,70] |
| Cytochrome C | Induces cytochrome C release into cytosol | [21,70,71] |
| Bcl-2 family | Increases Bax, Bad, Bax / Bcl2 ratio; decreases Bcl2, increases Bax / Bak translocation | [21,23,25,34,70] |
| AI-P | Induces expression of apoptose-inducing factor | [26] |
| Caspase 9 | Activates caspase 9 | [21,23,70,72,73] |
| Caspase 3 / 6 / 7 | Activates caspase 3 | [21,23,25,26,70,73] |
| Caspase 8 / 10 | Activates Caspase 8 / 10 via death receptors | [68,72] |
| PARP | Cleaves poly (ADP-ribose) polymerase | [21,26,70] |
| DFF-45 | Activates DNA fragmentation factor | [70] |
| Topoisomerase | Inhibits topoisomerase I and II | [60,61,64] |
| MAPK / ERK | Inhibits IGF induced activation of signaling; suppresses ERK expression | [21,36] |
| JNK / p38 | Activates JNK | [68,71] |
| p21 | Increases expression of p21 in p53 knockout cells | [36] |
| p21 | low doses induce, high doses suppress p21 expression | [23,33] |
| p53 | Stabilizes p53 via JNK activation; Accumulates p53 | [68] [24] |
| Cyclin | Inhibits expression of cyclin D1 induced by IGFR or pSTAT3 | [34,36] |
| survivin | Downregulates survivin | [23,34] |
| PCNA | Reduces expression of proliferating cell nuclear antigen | [21] |
| Fas/CD95 | Increases Fas/CD95 expression | [34] |
| TRAIL | Sensitizes TRAIL-induced apoptosis | [68] |
| XIAP | Reduces X-linked inhibitor of apoptosis protein | [74] |
| FASN | Inhibits fatty acid synthesis in cancer cells | [75] |
3.1 Inhibition of tumor cell proliferation
3.1.1 In vitro
3.1.2 In vivo
3.2 Anti-carcinogenic mechanisms
3.2.1 Protection from carcinogenic agents
3.2.2 Inhibition of cell adhesion and invasion
3.2.3 Topoisomerase inhibition
3.2.4 PI-3-Kinase / Akt regulation / MAPK / ERK / JNK
3.2.5 Mitochondrial activation of apoptosis
3.2.6 Death receptor-induced apoptosis and cell cycle arrestment mechanisms
3.2.7 Inhibition of fatty acid synthase (FASN)
3.2.8 Sensitization to chemotherapy
4. Epidemiological evidence for health protective effects of dietary flavone intake
5. Conclusions

References
- Block, G.; Patterson, B.; Subar, A. Fruit, vegetables, and cancer prevention: a review of the epidemiological evidence. Nutr. Cancer 1992, 18, 1–29. [Google Scholar]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food. Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Soobrattee, M.A.; Neergheen, V.S.; Luximon-Ramma, A.; Aruoma, O.I.; Bahorun, T. Phenolics as potential antioxidant therapeutic agents: mechanism and actions. Mutat. Res. 2005, 579, 2000–2013. [Google Scholar]
- Seelinger, G.; Merfort, I.; Schempp, C.M. Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin, a flavone from the Dyer’s weld Reseda luteola L. Planta Med. 2008. accepted. [Google Scholar]
- Brown, J.E.; Khodr, H.; Hider, R.C.; Rice-Evans, C.A. Structural dependence of flavonoid interactions with Cu++ ions: implications for their anti-oxidant properties. Biochem. J. 1998, 1173–1178. [Google Scholar]
- Heilmann, J.; Merfort, I. Aktueller Kenntnisstand zum Metabolismus von Flavonoiden. II. Resorption und Metabolismus von Flavonen, Flavanonen, Flavanen, Procyanidinen und Isoflavonoiden. Pharm. Uns. Zeit 1998, 27, 173–183. [Google Scholar] [CrossRef]
- Wittemer, S.M.; Ploch, M.; Windeck, T.; Müller, S.C.; Drewelow, B.; Derendorf, H.; Veit, M. Bioavailability and pharmacokinetics of caffeoylquinic acids and flavonoids after oral administration of Artichoke leaf extracts in humans. Phytomedicine 2005, 12, 28–38. [Google Scholar] [CrossRef]
- Boersma, MG.; van der Woude, H.; Bogaards, J.; Boeren, S.; Vervoort, J.; Cnubben, N.H.; van Iersel, M.L.; van Bladeren, P.J.; Rietjens, I.M. Regioselectivity of phase II metabolism of luteolin and quercetin by UDP-glucuronosyl transferases. Chem. Res. Toxicol. 2000, 15, 662–670. [Google Scholar]
- Shimoi, K.; Nakayama, T. Glucuronidase deconjugation in inflammation. Methods Enzymol. 2005, 400, 263–272. [Google Scholar]
- Merfort, I.; Heilmann, J; Hagedorn-Leweke, U.; Lippold, B.C. In vivo skin penetration studies on chamomile flavones. Pharmazie 1994, 49, 509–511. [Google Scholar]
- Chen, L.; Zhuang, H.-Y. Cancer preventive effects of the green tea polyphenol (-)-epigallocatechin-3-gallate. Molecules 2007, 12, 946–957. [Google Scholar] [CrossRef]
- Fotsis, T.; Pepper, M.S.; Aktas, E.; Breit, S.; Rasku, S.; Adlercreutz, H.; Wähälä, K.; Montesano, R.; Schweigerer, L. Flavonoids, dietary-derived inhibitors of cell proliferation and in vitro angiogenesis. Cancer Res. 1997, 57, 2916–2921. [Google Scholar]
- Kawaii, S.; Tomono, Y.; Katase, E.; Ogawa, K.; Yano, M. Antiproliferative activity of flavonoids on several cancer cell lines. Biosci. Biotechnol. Biochem. 1999, 63, 896–899. [Google Scholar] [CrossRef]
- Post, J.F.; Varma, R.S. Growth inhibitory effects of bioflavonoids and related compounds on human leukemic CEM-C1 and CEM-Cz cells. Cancer Lett. 1992, 67, 207–213. [Google Scholar] [CrossRef]
- Hirobe, Ch.; Quiao, Z.-S.; Takeya, K.; Itokawa, H. Cytotoxic flavonoids from Vitex agnus castus. Phytochemistry 1997, 46, 521–524. [Google Scholar] [CrossRef]
- Cherng, J.M.; Shieh, D.E.; Chiang, W.; Chang, M.Y.; Chiang, L.C. Chemopreventive effects of minor dietary constituents in common foods on human cancer cells. Biosci. Biotechnol. Biochem. 2007, 71, 1500–1504. [Google Scholar] [CrossRef]
- Takahashi, T.; Kobori, M.; Shinmoto, H.; Tsushida, T. Structure-activity relationships of flavonoids and the induction of granulocytic- or monocytic-differentiation in HL60 human myeloid leukemia cells. Biosci. Biotechnol. Biochem. 1998, 62, 2199–2204. [Google Scholar] [CrossRef]
- Ko, W.G.; Kang, T.H.; Lee, S.J.; Kim, Y.C.; Lee, B.H. Effects of luteolin on the inhibition of proliferation and induction of apoptosis in human myeloid leukaemia cells. Phytother. Res. 2002, 16, 295–298. [Google Scholar] [CrossRef]
- Chang, H.; Mi, M.T.; Gu, Y.Y.; Yuan, J.L.; Ling, W.H.; Lin, H. Effects of flavonoids with different structures on proliferation of leukemia cell line HL-60 [Article in Chinese]. Ai Zheng 2007, 26, 1309–1314. [Google Scholar]
- Huang, Y.T.; Hwang, J.J.; Lee, P.P.; Ke, F.C.; Huang, J.H.; Kandaswami, D.; Middleton, E., Jr.; Lee, M.T. Effects of luteolin and quercetin, inhibitors of tyrosine kinase, on cell growth and metastasis-associated properties in A 431 cells overexpressing epidermal growth factor receptor. Br. J. Pharmacol. 1999, 128, 999–1010. [Google Scholar]
- Kim, J.H.; Lee, E.O.; Lee, H.J.; Ku, J.S.; Lee, M.H.; Yang, D.C.; Kim, S.H. Caspase activation and extracellular signal-regulated kinase/Akt inhibition were involved in luteolin-induced apoptosis in Lewis lung carcinoma cells. Ann. N. Y. Acad. Sci. 2007, 1095, 598–611. [Google Scholar] [CrossRef]
- Knowles, L.M.; Zigrossi, D.A.; Tauber, R.A.; Hightower, C.; Milner, J.A. Flavonoids suppress androgen-independent human prostate tumor proliferation. Nutr Cancer 2000, 38, 116–122. [Google Scholar] [CrossRef]
- Lim, D.Y.; Jeong, Y.; Tyner, A.L.; Jung, H.Y.P. Induction of cell cycle arrest and apoptosis in HT-29 human colon cancer cells by the dietary compound luteolin. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G66–G75. [Google Scholar]
- Plaumann, B.; Fritsche, M.; Rimpler, H.; Brandner, G.; Hess, R.D. Flavonoids activate wild-type p53. Oncogene 1996, 13, 1605–1614. [Google Scholar]
- Chang, J.; Hsu, Y.; Kuo, P.; Kuo, Y.; Chiang, L.; Lin, C. Increase of Bax/ Bcl-XL ratio and arrest of cell cycle by luteolin in immortalized human hepatoma cell line. Life Sci. 2005, 76, 1883–1893. [Google Scholar] [CrossRef]
- Leung, H.W; Wu, C.H.; Lin, C.H.; Lee, H.Z. Luteolin induced DNA damage leading to human lung squamous carcinoma CH27 cell apoptosis. Eur. J. Pharmacol. 2005, 508, 77–83. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, J.C.; Chen, Y.; Huang, F.; Yang, M.S.; Xiao, P.G. Comparison of antioxidative and antitumor activities of six flavonoids from Epimedium koreanum [Article in Chinese]. Zhongguo Zhong Yao Za Zhi 2007, 32, 715–718. [Google Scholar]
- Pettit, G.R.; Hoard, M.S.; Doubek, D.L.; Schmidt, J.M.; Pettit, R.K.; Tackett, L.P.; Chapuis, J-Ch. The cancer cell growth inhibitory constituents of Terminalia arjuna (Combretaceae). J. Ethnopharmacol. 1996, 53, 57–63. [Google Scholar] [CrossRef]
- Leung, H.W.; Kuo, C.L.; Yang, W.H.; Lin, C.H.; Lee, H.Z. Antioxidant enzymes activity involvement in luteolin-induced human lung squamous carcinoma CH27 cell apoptosis. Eur. J. Pharmacol. 2006, 534, 12–18. [Google Scholar] [CrossRef]
- Makino, T.; Ono, T.; Muso, E.; Honsa, G. Inhibitory effect of Perilla frutescens and its phenolic constituents on cultured murine mesangial cell proliferation. Planta Med. 1998, 64, 541–545. [Google Scholar] [CrossRef]
- Mori, A.; Nishino, Ch.; Enoki, N.; Tawata, S. Cytotoxicity of plant flavonoids against HeLa cells. Phytochemistry 1988, 27, 1017–1020. [Google Scholar] [CrossRef]
- Woerdenbag, H.; Merfort, I.; Passreiter, C.M.; Schmidt, Th.J.; Willuhn, G.; van Uden, W.; Pras, N.; Kampinga, H.H.; Konings, A.W. Cytotoxicity of flavonoids and sesquiterpene lactones from arnica species against the GLC4 and the COLO 320 cell lines. Planta Med. 1994, 60, 434–437. [Google Scholar] [CrossRef]
- Chiang, C.T.; Way, T.D.; Lin, J.K. Sensitizing HER2-overexpressing cancer cells to luteolin-induced apoptosis through suppressing p21(WAF1/CIP1) expression with rapamycin. Mol. Cancer. Ther. 2007, 6, 2127–2138. [Google Scholar] [CrossRef]
- Selvendiran, K.; Koga, H.; Ueno, T.; Yoshida, T.; Maeyama, M.; Torimura, T.; Yano, H.; Kojiro, M.; Sata, M. Luteolin promotes degradation in signal transducer and activator of transcription 3 in human hepatoma cells: an implication for the antitumor potential of flavonoids. Cancer Res. 2006, 66, 4826–4834. [Google Scholar] [CrossRef]
- Chiu, F.L.; Lin, J.K. Downregulation of androgen receptor expression by luteolin causes inhibition of cell proliferation and induction of apoptosis in human prostate cancer cells and xenografts. Prostate 2008, 68, 61–71. [Google Scholar] [CrossRef]
- Fang, J.; Zhou, Q.; Shi, X.L.; Jiang, B.H. Luteolin inhibits insulin-like growth factor 1 receptor signaling in prostate cancer cells. Carcinogenesis 2007, 28, 713–723. [Google Scholar]
- Manju, V.; Nalini, N. Protective role of luteolin in 1,2-dimethylhydrazine induced experimental colon carcinogenesis. Cell Biochem. Funct. 2007, 25, 189–194. [Google Scholar] [CrossRef]
- Markaverich, B.M.; Alejandro, M.A. Bioflavonoids, type II [3H] estradiol binding sites and prostatic cancer cell proliferation. Int. J. Oncol. 1997, 11, 1311–1319. [Google Scholar]
- van der Bij, G.J.; Bögels, M.; Oosterling, S.J.; Kroon, J.; Schuckmann, D.T.; de Vries, H.E.; Meijer, S.; Beelen, R.H.; van Egmond, M. Tumor infiltrating macrophages reduce development of peritoneal colorectal carcinoma metastases. Cancer Lett. 2008. Jan 8 2008 [Epub ahead of print]. [Google Scholar]
- Ueda, H.; Yamazaki, C.; Yamazaki, M. Inhibitory effect of Perilla leaf extract and luteolin on mouse skin tumor promotion. Biol. Pharm. Bull. 2003, 26, 560–563. [Google Scholar] [CrossRef]
- Plaschke, K. Composition comprising one or more flavonoids, method of obtaining such composition and use thereof as UV-absorbing agent. US Pat. 6409996; B1,
- Gers-Barlag, H.; Kummerfeld, D.E.; Knüppel, A.; Müller, A.; Rümpel, D.E.; Stäb, F.; Dörschner, A.; Schönrock, U. Wirkstoffkombination aus sulfonierten UV-Filtersubstanzen und Flavonderivaten und/oder Flavanonderivaten, insbesondere Flanonoiden, sowie kosmetische Zubereitungen, solche Wirkstoffkombinationen enthaltend. Pat. Appl. DE 199 23 712 A 1 2000. [Google Scholar]
- Horváthová, K.; Novotný, L.; Vachálková, A. The free radical scavenging activity of four flavonoids determined by the comet assay. Neoplasma 2003, 50, 291–295. [Google Scholar]
- Horváthová, K.; Novotný, L.; Tóthová, D.; Vachálková, A. Determination of free radical scavenging activity of quercetin, rutin, luteolin and apigenin in H2O2-treated human ML cells K562. Neoplasma 2004, 51, 395–399. [Google Scholar]
- Horváthová, K.; Chalupa, L.; Sebova, D.; Tóthová, D.; Vachálková, A. Protective effect of quercetin and luteolin in human melanoma HMB-2 cells. Mut. Res. 2005, 565, 105–112. [Google Scholar] [CrossRef]
- Noroozi, M.; Angerson, W.J.; Lean, M.E. Effects of flavonoids and vitamin C on oxidative DNA damage to human lymphocytes. Am. J. Nutr. 1998, 67, 1210–1218. [Google Scholar]
- Steffan, B. Inhaltsstoffe aus Pflanzen der indonesischen Volksmedizin (Jamu): Isolierung, Identifizierung, und Charakterisierung der antioxidativen Eigenschaften. Doctoral thesis, 2005. [Google Scholar]
- Shimoi, K.; Masuda, S.; Shen, B.; Furugori, M.; Kinae, N. Radioprotective effect of antioxidative flavonoids in gamma-ray irradiated mice. Carcinogenesis 1994, 15, 2669–2672. [Google Scholar] [CrossRef]
- Sharma, V.; Mishra, M.; Ghosh, S.; Tewari, R.; Basu, A.; Seth, P.; Sen, E. Modulation of interleukin-1beta mediated inflammatory response in human astrocytes by flavonoids: implications in neuroprotection. Brain Res. Bull. 2007, 73, 55–63. [Google Scholar] [CrossRef]
- Samy, R.P.; Gopalakrishnakone, P.; Ignacimuthu, S. Anti-tumor promoting potential of luteolin against 7,12-dimethylbenz(a)anthracene-induced mammary tumors in rats. Chem. Biol. Interact. 2006, 164, 1–14. [Google Scholar] [CrossRef]
- Manju, V.; Nalini, N. Chemopreventive potential of luteolin during colon carcinogenesis induced by 1,2-dimethylhydrazine. Ital. J. Biochem. 2005, 54, 268–275. [Google Scholar]
- Choi, J.S.; Park, K.Y.; Moon, S.H.; Rhee, S.H.; Young, H.S. Antimutagenic effect of plant flavonoids in the Salmonella assay system. Arch. Pharm. Res. 1994, 17, 71–75. [Google Scholar] [CrossRef]
- Taj, S.; Nagarajan, B. Inhibition by quercetin and luteolin of chromosomal alterations induced by salted, deep-fried fish and mutton in rats. Mutat. Res. 1996, 369, 97–106. [Google Scholar] [CrossRef]
- Li, Y.C.; Hung, C.F.; Yeh, F.T.; Lin, J.P.; Chung, J.G. Luteolin-inhibited arylamine N-acetyltransferase activity and DNA-2-aminofluorene adduct in human and mouse leukemia cells. Food Chem. Toxicol. 2001, 39, 641–647. [Google Scholar] [CrossRef]
- Lu, K.H.; Lin, K.L.; Yang, C.C.; Hsia, T.C.; Hsiao, Y.M.; Chou, M.C.; Ho, H.C.; Chung, J.G. The effect of paclitaxel on gene expression and activity of arylamine N-acetyltransferase and DNA-2-aminofluorene adduct formation in human leukemia HL-60 cells. Food Chem. Toxicol. 2002, 40, 705–713. [Google Scholar] [CrossRef]
- Chen, J.C.; Chung, J.G.; Lin, K.M. Effects of luteolin on arylamine N-acetyltransferase activity in human liver tumour cells. Cytobios 2000, 102, 95–106. [Google Scholar]
- Su, C.C.; Chen, G.W.; Yeh, C.C.; Yang, M.D.; Hung, C.F.; Chung, J.G. Luteolin induces N-acetylation and DNA adduct of 2-aminofluorene accompanying N-acetyltransferase activity and gene expression in human bladder cancer T24 cell line. Anticancer Res. 2003, 23, 355–362. [Google Scholar]
- Lee, W.J.; Wu, L.F.; Chen, W.K.; Wang, C.J.; Tseng, T.H. Inhibitory effect of luteolin on hepatocyte growth factor/scatter factor-induced HepG2 cell invasion involving both MAPK/ERKs and PI3K-Akt pathways. Chem. Biol. Interact. 2006, 160, 123–133. [Google Scholar] [CrossRef]
- Lansky, E.P.; Harrison, G.; Froom, P.; Jiang, W.G. Pomegranate (Punica granatum) pure chemicals show possible synergistic inhibition of human PC-3 prostate cancer cell invasion across Matrigel. Invest. New Drugs 2005, 23, 121–122. [Google Scholar] [CrossRef]
- Yamashita, N.; Kawanishi, S. Distinct mechanisms of DNA damage in apoptosis induced by quercetin and luteolin. Free Radic. Res. 2000, 33, 623–633. [Google Scholar] [CrossRef]
- Cantero, G.; Campanella, C.; Mateos, S.; Cortés, F. Topoisomerase II inhibition and high yield of endoreduplication induced by the flavonoids luteolin and quercetin. Mutagenesis 2006, 21, 321–325. [Google Scholar] [CrossRef]
- Sugimura, T.; Nagao, M.; Matsushima, T.; et al. Mutagenicity of flavone derivatives. Proc. Jpn. Acad. 1977, 53, 194–197. [Google Scholar] [CrossRef]
- van der Hoeven, J.C.M.; Bruggeman, I.M.; Debets, F.M.H. Genotoxicity of quercetin in cultured mammalian cells. Mutat. Res. 1984, 136, 9–12. [Google Scholar] [CrossRef]
- Chowdhury, A.R.; Sharma, S.; Mandal, S.; Goswami, A.; Mukhopadhyay, S.; Majumder, H.K. Luteolin, an emerging anti-cancer flavonoid, poisons eukaryotic DNA topoisomerase. Biochem. J. 2002, 366, 653–661. [Google Scholar] [CrossRef]
- Ruiz, P.A.; Haller, D. Functional diversity of flavonoids in the inhibition of the proinflammatory NF-kappaB, IRF, and Akt signaling pathways in murine intestinal epithelial cells. J. Nutr. 2006, 136, 664–671. [Google Scholar]
- Agullo, G.; Gamet-Payrastre, L.; Manenti, S.; Viala, C.; Rémésy, C.; Chap, H.; Payrastre, B. Relationship between flavonoid structure and inhibition of phosphatidylinositol 3-kinase: a comparison with tyrosine kinase and protein kinase C inhibition. Biochem. Pharmacol. 1997, 53, 1649–1657. [Google Scholar] [CrossRef]
- Liu, W.; Liang, N.C.; Huang, R.B. Effect of three flavones on enzyme activity of recombinant human phosphoinositide 3-kinase p110beta catalytic subunit [Article in Chinese]. Zhong Yao Cai 2006, 29, 33–36. [Google Scholar]
- Shi, R.; Huang, Q.; Zhu, X.; Ong, Y.B.; Zhao, B.; Lu, J.; Ong, C.N.; Shen, H.M. Luteolin sensitizes the anticancer effect of cisplatin via c-Jun NH2-terminal kinase-mediated p53 phosphorylation and stabilization. Mol. Cancer Ther. 2007, 6, 1338–1347. [Google Scholar] [CrossRef]
- Han, H.Y.; Shan, S.; Zhang, X.; Wang, N.L.; Lu, X.P.; Yao, X.S. Down-regulation of prostate specific antigen in LNCaP cells by flavonoids from the pollen of Brassica napus L. Phytomedicine 2007, 14, 338–343. [Google Scholar] [CrossRef]
- Cheng, A.C.; Huang, T.C.; Lai, C.S.; Pan, M.H. Induction of apoptosis by luteolin through cleavage of Bcl-2 family in human leukemia HL-60 cells. Eur. J. Pharmacol. 2005, 509, 1–10. [Google Scholar] [CrossRef]
- Lee, H.J.; Wang, C.J.; Kuo, H.C.; Chou, F.P.; Jean, L.F.; Tseng, T.H. Induction apoptosis of luteolin in human hepatoma HepG2 cells involving mitochondria translocation of Bax/Bak and activation of JNK. Toxicol. Appl. Pharmacol. 2005, 203, 124–131. [Google Scholar] [CrossRef]
- Horinaka, M.; Yoshida, T.; Shiraishi, T.; Nakata, S.; Wakada, M.; Nakanishi, R.; Nishino, H.; Matsui, H.; Sakai, T. Luteolin induces apoptosis via death receptor 5 upregulation in human malignant tumor cells. Oncogene 2005, 24, 7180–7189. [Google Scholar] [CrossRef]
- Michels, G.; Wätjen, W.; Niering, P.; Steffan, B.; Thi, Q.H.; Chovolou, Y.; Kampkötter, A.; Bast, A.; Proksch, P.; Kahl, R. Pro-apoptotic effects of the flavonoid luteolin in rat H4IIE cells. Toxicology 2005, 206, 337–348. [Google Scholar] [CrossRef]
- Shi, R.X.; Ong, C.N.; Shen, H.M. Protein kinase C inhibition and x-linked inhibitor of apoptosis protein degradation contribute to the sensitization effect of luteolin on tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in cancer cells. Cancer Res. 2005, 65, 7815–7823. [Google Scholar]
- Lupu, R.; Menendez, J.A. Pharmacological inhibitors of Fatty Acid Synthase (FASN)--catalyzed endogenous fatty acid biogenesis: a new family of anti-cancer agents? Curr. Pharm. Biotechnol. 2006, 7, 483–493. [Google Scholar] [CrossRef]
- van Zanden, J.J.; Geraets, L.; Wortelboer, H.M.; van Bladeren, P.J.; Rietjens, I.M.C.M.; Cnubben, N.H.P. Structural requirements for the flavonoid-mediated modulation of glutathione S-transferase P1-1 and GS-X pump activity in MCF7 breast cancer cells. Biochem. Pharmacol. 2004, 67, 1607–1617. [Google Scholar] [CrossRef]
- Graefe, E.U.; Derendorf, H.; Veit, M. Pharmacokinetics and bioavailability of the flavonols quercetin in humans. Int. J. Clin. Pharmacol. Ther. 1999, 37, 219–233. [Google Scholar]
- Anonymous. USDA Databank for the flavonoid content of selected foods, 2nd release. 2007. Available online: http://www.ars.usda.gov/nutrientdata.
- Bosetti, C.; Spertini, L.; Parpinel, M.; Gnagnarella, P.; Lagiou, P.; Negri, E.; Franceschi, S.; Montella, M.; Peterson, J.; Dwyer, J.; Giacosa, A.; La Vecchia, C. Flavonoids and breast cancer risk in Italy. Cancer Epidemiol. Biomarkers Prev. 2005, 14, 805–808. [Google Scholar] [CrossRef]
- Peterson, J.; Lagiou, P.; Samoli, E.; Lagiou, A.; Katsouyanni, K.; La Vecchia, C.; Dwyer, J.; Trichopoulos, D. Flavonoid intake and breast cancer risk: a case--control study in Greece. Br. J. Cancer 2003, 89, 1255–1259. [Google Scholar] [CrossRef]
- Fink, B.N.; Gaudet, M.M.; Britton, J.A.; Abrahamson, P.E.; Teitelbaum, S.L.; Jacobson, J.; Bell, P.; Thomas, J.A; Kabat, G.C.; Neugut, A.I.; Gammon, M.D. Fruits, vegetables, and micronutrient intake in relation to breast cancer survival. Breast Cancer Res. Treat. 2006, 98, 199–208. [Google Scholar] [CrossRef]
- Fink, B.N.; Steck, S.E.; Wolff, M.S.; Britton, J.A.; Kabat, G.C.; Gaudet, M.M.; Abrahamson, P.E.; Bell, P.; Schroeder, J.C.; Teitelbaum, S.L.; Neugut, A.I.; Gammon, M.D. Dietary Flavonoid Intake and Breast Cancer Survival among Women on Long Island. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 2285–2292. [Google Scholar] [CrossRef]
- Rossi, M.; Negri, E.; Talamini, R.; Bosetti, C.; Parpinel, M.; Gnagnarella, P.; Franceschi, S.; Dal Maso, L.; Montella, M.; Giacosa, A.; La Vecchia, C. Flavonoids and Colorectal Cancer in Italy. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 1555–1558. [Google Scholar] [CrossRef]
- Bosetti, C.; Rossi, M.; McLaughlin, JK; Negri, E.; Talamini, R.; Lagiou, P.; Montella, M.; Ramazzotti, V.; Franceschi, S.; LaVecchia, C. Flavonoids and the risk of renal cell carcinoma. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 98–101. [Google Scholar] [CrossRef]
- Garcia-Closas, R.; Agudo, A.; Gonzalez, C.A.; Riboli, E. Intake of specific carotenoids and flavonoids and the risk of lung cancer in women in Barcelona, Spain. Nutr. Cancer 1998, 32, 154–158. [Google Scholar] [CrossRef]
- Neuhouser, M.L. Dietary flavonoids and cancer risk: evidence from human population studies. Nutr. Cancer. 2004, 50, 1–7. [Google Scholar] [CrossRef]
- Gates, M.A.; Tworoger, S.S.; Hecht, J.L.; De Vivo, I.; Rosner, B.; Hankinson, S.E. A prospective study of dietary flavonoid intake and incidence of epithelial ovarian cancer. Int. J. Cancer 2007, 121, 2225–2232. [Google Scholar] [CrossRef]
- Sample Availability: not available.
© 2008 by the authors. Licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Seelinger, G.; Merfort, I.; Wölfle, U.; Schempp, C.M. Anti-carcinogenic Effects of the Flavonoid Luteolin. Molecules 2008, 13, 2628-2651. https://doi.org/10.3390/molecules13102628
Seelinger G, Merfort I, Wölfle U, Schempp CM. Anti-carcinogenic Effects of the Flavonoid Luteolin. Molecules. 2008; 13(10):2628-2651. https://doi.org/10.3390/molecules13102628
Chicago/Turabian StyleSeelinger, Günter, Irmgard Merfort, Ute Wölfle, and Christoph M. Schempp. 2008. "Anti-carcinogenic Effects of the Flavonoid Luteolin" Molecules 13, no. 10: 2628-2651. https://doi.org/10.3390/molecules13102628
APA StyleSeelinger, G., Merfort, I., Wölfle, U., & Schempp, C. M. (2008). Anti-carcinogenic Effects of the Flavonoid Luteolin. Molecules, 13(10), 2628-2651. https://doi.org/10.3390/molecules13102628