Formation of Coatings Based on Titanium Dioxide Nanosolson Polyester Fibre Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Synthesis of Nanoparticles
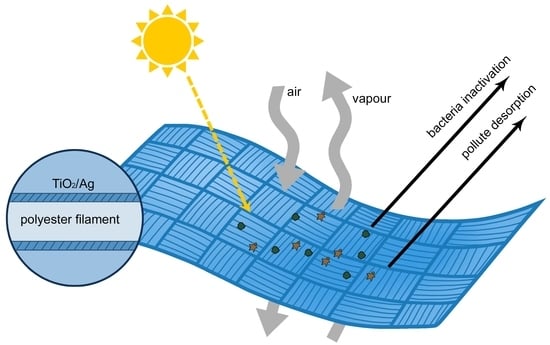
2.2.2. Polyester Fabric Preparation and Modification
2.2.3. Testing of Modified Polyester Fabric
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mahltig, B.; Textor, T. Nanosols and Textiles; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2008; p. 224. [Google Scholar]
- Gashti, M.P.; Pakdel, E.; Alimohammadi, F. Nanotechnology-based coating techniques for smart textiles. In Active Coatings for Smart Textiles; Woodhead Publishing: Cambridge, UK, 2016; pp. 243–268. [Google Scholar]
- Nurhan, O.C.; Buket, A. Sol-Gel Applications in Textile Finishing Processes. In Recent Applications in Sol-Gel Synthesis; INTECH: London, UK, 2017; pp. 253–281. [Google Scholar]
- Ismail, W.N.W. Sol–gel technology for innovative fabric finishing—A review. J. Sol-Gel Sci. Technol. 2016, 78, 698–707. [Google Scholar] [CrossRef] [Green Version]
- Rivero, P.J.; Urrutia, A.; Goicoechea, J.; Arregui, F.J. Nanomaterials for Functional Textiles and Fibers. Nanoscale Res. Lett. 2015, 10, 501–523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galrajani, M.L.; Gupta, D. Emerging techniques for functional finishing of textiles. IJFTR 2011, 36, 388–397. [Google Scholar]
- Schmidt, H. Considerations about the sol-gel process: From the classical sol-gel route to advanced chemical nanotechnologies. J. Sol-Gel Sci. Technol. 2006, 40, 115–130. [Google Scholar] [CrossRef]
- Hench, L.L.; West, J.K. The Sol-Gel Process. Chem. Rev. 1990, 90, 33–72. [Google Scholar] [CrossRef]
- Brinker, C.J.; Scherer, G. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing; Academic Press: Boston, MA, USA, 1990; p. 908. [Google Scholar]
- Li, S.; Huang, J.; Chen, Z.; Chena, G.; Lai, Y. A review on special wettability textiles: Theoretical models, fabrication technologies and multifunctional applications. J. Mater. Chem. A 2017, 5, 31–55. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.C.; Wang, C.C. Crosslinking of cotton cellulose with succinic acid in the presence of titanium dioxide nano-catalyst under UV irradiation. J. Sol-Gel Sci. Technol. 2006, 40, 31–38. [Google Scholar] [CrossRef]
- Agafonov, V.V.; Vinogradov, A.V. A new approach to apply crystalline titania hydrosols onto a polyester cloth. Mendeleev Commun. 2013, 23, 286–288. [Google Scholar] [CrossRef]
- Bozzi, A.; Yuranova, T.; Kiwi, J. Self-cleaning of wool-polyamide and polyester textiles by TiO2-rutile modification under daylight irradiation at ambient temperature. J. Photochem. Photobiol. A 2005, 172, 27–34. [Google Scholar] [CrossRef]
- Yuranova, T.; Laub, D.; Kiwi, J. Synthesis, activity and characterization of textiles showing self-cleaning activity under daylight irradiation. Catal. Today 2007, 122, 109–117. [Google Scholar] [CrossRef]
- Galoppini, E. Linkers for anchoring sensitizers to semiconductor nanoparticles. Coord. Chem. Rev. 2004, 248, 1283–1297. [Google Scholar] [CrossRef]
- Huang, K.; Yang, K.; Lin, S.; Lian, W. Antiwrinkle treatment of cotton fabric with a mixed sol of TEOS-TTB/DMDHEU. J. Appl. Polym. Sci. 2007, 106, 2559–2564. [Google Scholar] [CrossRef]
- Galkina, O.L.; Vinogradov, V.V.; Vinogradov, V.V.; Agafonov, A.V. Development of the temperature sol-gel synthesis of TiO2 provide self-cleaning effect on the textile materials. Nanotechnol. Russ. 2012, 7, 604–614. [Google Scholar] [CrossRef]
- Cerveau, G.; Corriu, R.J.P.; Lerouge, F.; Bellec, N.; Lorcy, D.; Nobili, M. Self-organization of a tetrasubstitutedtetrathiafulvalene in a silica based hybrid organic-inorganic material. Chem. Commun. 2004, 4, 396–397. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Kumeeva, T.Y.; Agafonov, A.V.; Ivanov, V.K. Modification of polyester fabrics with nanosized titanium dioxide to impart photoactivity. Inorg. Mater. Appl. Res. 2017, 8, 696–703. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Vavilova, S.Y. The modifying effect of ammonium nitrate and sodium hydroxide on poly(ethylene terephthalate) materials. Fiber Chem. 2004, 36, 413–415. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Chorev, A.V.; Vavilova, S.Y. Chemical method of surface activation of poly(ethylene terephthalate) fibre materials. Part 1. Study of the modifying effect of sodium hydroxide solution and products made from quaternary ammonium salts. Fiber Chem. 2009, 41, 158–163. [Google Scholar] [CrossRef]
- Kuzmin, S.M.; Prorokova, N.P.; Khorev, A.V. Chapter 19. Plasma-assisted modification of textile yarns in liquid environment. In Textiles: Types, Uses and Production Methoda; El Nemr, A., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2012; pp. 557–578. [Google Scholar]
- Prorokova, N.P.; Chorev, A.V.; Kuzmin, S.M.; Vavilova, S.Y.; Prorokov, V.N. Chemical Method of Fibrous Materials Surface Activation on the Basis of Polyethylene Terephthalate (PET). Chem. Chem. Technol. 2014, 8, 293–302. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Kumeeva, T.Y.; Kuz’min, S.M.; Kholodkov, I.V. Modification of polyester fibrous materials with surface barrier discharge for making them more hydrophilic. Russ. J. Appl. 2016, 89, 111–118. [Google Scholar] [CrossRef]
- Prasad, G.K.; Agarwal, G.S.; Singh, B.; Rai, G.P.; Vijayaraghavan, R. Photocatalytic inactivation of Bacillus anthracis by titania nanomaterials. J. Hazard. Mater. 2009, 165, 506–510. [Google Scholar] [CrossRef]
- Linsebigler, A.L.; Lu, G.; Yates, J.T., Jr. Photocatalysis on TiO2 Surfaces: Principles, Mechanisms, and Selected Results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Neri, G.; Rizzo, G.; Galvagno, S.; Loiacono, G.; Donato, A.; Musolino, M.G.; Pietropaolo, R.; Rombi, E. Sol-gel synthesis, characterization and catalytic properties of Fe-Ti mixed oxides. Appl. Catal. A Gen. 2004, 274, 243–251. [Google Scholar] [CrossRef]
- Pal, B.; Hata, T.; Goto, K.; Nogami, S. Photocatalytic degradation of o-cresol sensitized by iron-titania binary photocatalysts. J. Mol. Catal. A Chem. 2001, 169, 147–155. [Google Scholar] [CrossRef]
- Wang, C.-T.; Ro, S.-H. Nanoparticle iron-titanium oxide aerogels. Mater. Chem. Phys. 2007, 101, 41–48. [Google Scholar] [CrossRef]
- Lei, X.F.; Xue, X.X.; Yang, H. Preparation and characterization of Ag-doped TiO2 nanomaterials and their photocatalytic reduction of Cr(VI) under visible light. Appl. Surf. Sci. 2014, 321, 396–403. [Google Scholar] [CrossRef]
- Sokmen, M.; Candan, F.; Sumer, Z. Disinfection of E. coli by the Ag–TiO2/UV system: Lipidperoxidation. Photochem. Photobiol. A Chem. 2001, 143, 241–244. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.L.; Yang, F.; Yang, X.R. Excellent antimicrobial properties of mesoporous anatase TiO2 and Ag/TiO2 composite films. Micropor. Mesopor. Mater. 2008, 114, 431–439. [Google Scholar] [CrossRef]
- Apphouse, P.; Varghese, A.; Tendero, C. Stable hydrosols for TiO2 coatings. J. Sol-Gel Sci. Technol. 2010, 56, 250–263. [Google Scholar] [CrossRef] [Green Version]
- Prorokova, N.P.; Kumeeva, T.Y.; Gerasimova, T.V.; Agafonov, A.V. Effect of the structure of Fe-doped titania-based nanocomposites on the photocatalytic activity of polyester fabrics modified by them. Inorg. Mater. 2017, 53, 1336–1342. [Google Scholar] [CrossRef]
- Agafonov, A.V.; Vinogradov, A.V. Sol-gel synthesis, preparation and characterization of photoactive TiO2 with ultrasound treatment. J. Sol-Gel Sci. Technol. 2009, 49, 180–185. [Google Scholar] [CrossRef]
- Khalyavka, T.A.; Shimanovskaya, V.V.; Strelko, V.V.; Kapinus, E.I. Photocatalytic activity of titanium dioxide in the degradation of methylene blue and tetrachlorofluorescein in aqueous solutions. Theor. Exp. Chem. 2001, 37, 58–62. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Kumeeva, T.Y.; Novikov, V.V.; Holodkov, I.V. Regulation of the tribological characteristics of polyester fabrics by surface modification using tetrafluoroethylene telomeres. J. Frict. Wear 2018, 39, 121–128. [Google Scholar] [CrossRef]
- ASTM E2149-10 Standard Test Method for Determining the Antimicrobial Activity of Immobilized Antimicrobial Agents under Dynamic Contact Conditions; ASTM International: West Conshohochen, PA, USA, 2001.
- Bui, T.H.; Felix, C.; Pigeot-Remy, S.; Herrmann, J.M.; Lejeune, P.; Guillard, C. Photocatalytic inactivation of wild and hyper-adherent Escherichia coli strains in presence of suspended or supported TiO2: Influence of the Isoelectric point of the particle size and of the adsorptive properties of titania. J. Adv. Oxid. Technol. 2008, 11, 510–518. [Google Scholar] [CrossRef]
- Nadtochenko, V.A.; Radtsig, M.A.; Khmel, I.A. Antimicrobial effect of metallic and semiconductor nanoparticles. Nanotechnol. Russ. 2010, 5, 277–289. [Google Scholar] [CrossRef]
- Zolotavin, P.; Permenova, E.; Sarkisov, O.; Nadtochenko, V.; Azouani, R.; Portes, P.; Chhor, K.; Kanaev, A. Two-photon luminescence enhancement of silver nanoclusters photodeposited onto mesoporous TiO2 film. Chem. Phys. Lett. 2008, 457, 342–346. [Google Scholar] [CrossRef]
- Zhang, H.J.; Chen, G.H. Potent antibacterial activities of Ag/TiO2 nanocomposite powders synthesized by a one-pot sol-gel method. Environ. Sci. Technol. 2009, 43, 2905–2910. [Google Scholar] [CrossRef] [PubMed]
- Prorokova, N.P.; Kumeeva, T.Y.; Kuznetsov, O.Y. Antimicrobial properties of polyester fabric modified by nanosized titanium dioxide. Inorg. Mater. Appl. Res. 2018, 9, 250–256. [Google Scholar] [CrossRef]
- Wang, R.; Wang, Y. Characterization of titania hydrosol and its influence on light fastness of polyester fabric. Procedia Eng. 2011, 18, 307–311. [Google Scholar] [CrossRef] [Green Version]
- Selishev, D.S.; Karaseva, I.P.; Uvaev, V.V.; Kozlov, D.V.; Parmon, V.N. Effect of preparation method of functionalized textile materials on their photocatalytic activity and stability under UV irradiation. Chem. Eng. J. 2013, 224, 114–120. [Google Scholar] [CrossRef]







| Composition of the Modified Polyester Fabric Coating | Stiffness of PEF Fabric |
|---|---|
| Uncoated Fabric | 1 |
| TiO2 nanoparticles, 4 g/m2 | 1.27 |
| TiO2 nanoparticles, 8 g/m2 | 1.53 |
| TiO2 nanoparticles, 16 g/m2 | 2.00 |
| Composition of the Modified Polyester Fabric Coating | Probe Adhesion to the Coating, nN | Relative Stiffness, nA/nm | ||
|---|---|---|---|---|
| Initial Sample | Sample Subjected to Abrasion | Initial Sample | Sample Subjected to Abrasion | |
| TiO2 undoped nanoparticles | 11.6 | 7.7 | 0.052 | 0.045 |
| TiO2 nanoparticles doped with iron | 16.8 | 21.0 | 0.058 | 0.054 |
| TiO2 nanoparticles doped with silver | 19.8 | 15.2 | 0.053 | 0.049 |
| Composition of the Modified Polyester Fabric Coating | Growth (+) or Inhibition (−) of Pathogenic Cultures, % | ||
|---|---|---|---|
| E. coli | S. aureus | C. albicans | |
| TiO2 nanoparticles | +31 | +63 | +83 |
| TiO2 nanoparticles doped with iron | −27 | +53 | +61 |
| TiO2 nanoparticles doped with silver | −50 | +35 | +8 |
| Type of Polyester Fabric | Specific Tensile Strength, MPa | Relative Breaking Elongation, % |
|---|---|---|
| Untreated | 1410 ± 46 | 26.7 ± 4.0 |
| Activated with 0.375 mol/L NaOH | 1318 ± 45 | 25.6 ± 1.2 |
| Untreated, after 250 min UV-exposure | 1381 ± 67 | 27.0 ± 1.6 |
| With a TiO2-based coating after NaOH activation | 1507 ± 64 | 23.8 ± 1.2 |
| With a TiO2-based coating after NaOH activation and 250 min UV-exposure | 1460 ± 57 | 23.8 ± 1.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prorokova, N.; Kumeeva, T.; Kholodkov, I. Formation of Coatings Based on Titanium Dioxide Nanosolson Polyester Fibre Materials. Coatings 2020, 10, 82. https://doi.org/10.3390/coatings10010082
Prorokova N, Kumeeva T, Kholodkov I. Formation of Coatings Based on Titanium Dioxide Nanosolson Polyester Fibre Materials. Coatings. 2020; 10(1):82. https://doi.org/10.3390/coatings10010082
Chicago/Turabian StyleProrokova, Natalia, Tatiana Kumeeva, and Igor Kholodkov. 2020. "Formation of Coatings Based on Titanium Dioxide Nanosolson Polyester Fibre Materials" Coatings 10, no. 1: 82. https://doi.org/10.3390/coatings10010082
APA StyleProrokova, N., Kumeeva, T., & Kholodkov, I. (2020). Formation of Coatings Based on Titanium Dioxide Nanosolson Polyester Fibre Materials. Coatings, 10(1), 82. https://doi.org/10.3390/coatings10010082