Potentials of Neuropeptides as Therapeutic Agents for Neurological Diseases
Abstract
:1. Introduction
2. Function of Neuropeptides in the Neurological System
2.1. Neuropeptidergic Modulation of Neurodevelopment
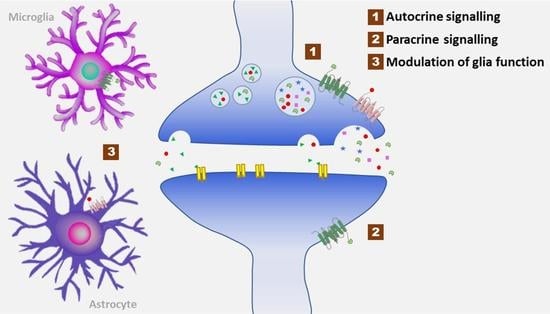
2.2. Involvement of Neuropeptides in Normal Neurological Homeostasis
2.3. Neuropeptides and Sensory Perception
2.4. Neuropeptides and the Systemic Inflammatory Response
3. Interrelation between Neuropeptide Signalling, Metabolic Dysregulation, and Neurological Dysfunction
4. Potential of Neuropeptides in Resolving Outstanding Questions in Neurological Research
5. Synthetic Approach, Strategies, and Prospects for the Development of Peptide-Based Neurotherapeutics
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mensah, G.A.; Wei, G.S.; Sorlie, P.D.; Fine, L.J.; Rosenberg, Y.; Kaufmann, P.G.; Mussolino, M.E.; Hsu, L.L.; Addou, E.; Engelgau, M.M.; et al. Decline in Cardiovascular Mortality: Possible Causes and Implications. Circ. Res. 2017, 120, 366–380. [Google Scholar] [CrossRef] [PubMed]
- Lv, S.; Wang, Y.; Sun, T.; Wan, D.; Sheng, L.; Li, W.; Zhu, H.; Li, Y.; Lu, J. Overall Survival Benefit from Trastuzumab-Based Treatment in HER2-Positive Metastatic Breast Cancer: A Retrospective Analysis. Oncol. Res. Treat. 2018, 41, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Maude, S.L.; Frey, N.; Shaw, P.A.; Aplenc, R.; Barrett, D.M.; Bunin, N.J.; Chew, A.; Gonzalez, V.E.; Zheng, Z.; Lacey, S.F.; et al. Chimeric Antigen Receptor T Cells for Sustained Remissions in Leukemia. N. Engl. J. Med. 2014, 371, 1507–1517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbasi, J. COVID-19 mRNA Vaccines Blunt Breakthrough Infection Severity. JAMA 2021, 326, 473. [Google Scholar] [CrossRef]
- Bespalov, A.; Steckler, T.; Altevogt, B.; Koustova, E.; Skolnick, P.; Deaver, D.; Millan, M.J.; Bastlund, J.F.; Doller, D.; Witkin, J.; et al. Failed trials for central nervous system disorders do not necessarily invalidate preclinical models and drug targets. Nat. Rev. Drug Discov. 2016, 15, 516. [Google Scholar] [CrossRef] [Green Version]
- Clarfield, A.M. The Decreasing Prevalence of Reversible Dementias: An Updated Meta-analysis. Arch. Intern. Med. 2003, 163, 2219. [Google Scholar] [CrossRef] [Green Version]
- Dutta, R.; Trapp, B.D. Mechanisms of neuronal dysfunction and degeneration in multiple sclerosis. Prog. Neurobiol. 2011, 93, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Pardridge, W.M. Blood-Brain Barrier and Delivery of Protein and Gene Therapeutics to Brain. Front. Aging Neurosci. 2020, 11, 373. [Google Scholar] [CrossRef]
- Groß, A.; Hashimoto, C.; Sticht, H.; Eichler, J. Synthetic Peptides as Protein Mimics. Front. Bioeng. Biotechnol. 2016, 3, 211. [Google Scholar] [CrossRef] [Green Version]
- Lau, J.L.; Dunn, M.K. Therapeutic peptides: Historical perspectives, current development trends, and future directions. Bioorg. Med. Chem. 2018, 26, 2700–2707. [Google Scholar] [CrossRef]
- Muttenthaler, M.; King, G.F.; Adams, D.J.; Alewood, P.F. Trends in peptide drug discovery. Nat. Rev. Drug Discov. 2021, 20, 309–325. [Google Scholar] [CrossRef]
- Usmani, S.S.; Bedi, G.; Samuel, J.S.; Singh, S.; Kalra, S.; Kumar, P.; Ahuja, A.A.; Sharma, M.; Gautam, A.; Raghava, G.P.S. THPdb: Database of FDA-approved peptide and protein therapeutics. PLoS ONE 2017, 12, e0181748. [Google Scholar] [CrossRef] [Green Version]
- Kim, R.J.; Malattia, C.; Allen, M.; Moshang, T.; Maghnie, M. Vasopressin and desmopressin in central diabetes insipidus: Adverse effects and clinical considerations. Pediatr. Endocrinol. Rev. PER 2004, 2 (Suppl. 1), 115–123. [Google Scholar]
- Fralick, M.; Kesselheim, A.S. FDA Approval of Desmopressin for Nocturia. JAMA 2017, 317, 2059. [Google Scholar] [CrossRef]
- Valenti, G.; Tamma, G. History of Diabetes Insipidus. G. Ital. Nefrol. Organo Uff. Della Soc. Ital. Nefrol. 2016, 33 (Suppl. 66), 33S66. [Google Scholar]
- Page, K.; McCool, W.F.; Guidera, M. Examination of the Pharmacology of Oxytocin and Clinical Guidelines for Use in Labor. J. Midwifery Womens Health 2017, 62, 425–433. [Google Scholar] [CrossRef]
- Du Vigneaud, V.; Ressler, C.; Swan, J.M.; Roberts, C.W.; Katsoyannis, P.G. The Synthesis of Oxytocin 1. J. Am. Chem. Soc. 1954, 76, 3115–3121. [Google Scholar] [CrossRef]
- Hirsch, I.B. Insulin Analogues. N. Engl. J. Med. 2005, 352, 174–183. [Google Scholar] [CrossRef]
- Havelund, S.; Plum, A.; Ribel, U.; Jonassen, I.; Vølund, A.; Markussen, J.; Kurtzhals, P. The Mechanism of Protraction of Insulin Detemir, a Long-Acting, Acylated Analog of Human Insulin. Pharm. Res. 2004, 21, 1498–1504. [Google Scholar] [CrossRef]
- Ryberg, L.A.; Sønderby, P.; Barrientos, F.; Bukrinski, J.T.; Peters, G.H.J.; Harris, P. Solution structures of long-acting insulin analogues and their complexes with albumin. Acta Crystallogr. Sect. Struct. Biol. 2019, 75, 272–282. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, Y.B.; Kim, B.H.; Lee, C.; Cho, Y.M.; Kim, S.-N.; Park, C.G.; Cho, Y.-C.; Choy, Y.B. Implantable batteryless device for on-demand and pulsatile insulin administration. Nat. Commun. 2017, 8, 15032. [Google Scholar] [CrossRef] [Green Version]
- Kong, X.; Luo, M.; Cai, L.; Zhang, P.; Yan, R.; Hu, Y.; Li, H.; Ma, J. Needle-free jet injection of insulin glargine improves glycemic control in patients with type 2 diabetes mellitus: A study based on the flash glucose monitoring system. Expert Opin. Drug Deliv. 2021, 18, 635–641. [Google Scholar] [CrossRef]
- Ahad, A.; Raish, M.; Bin Jardan, Y.A.; Al-Mohizea, A.M.; Al-Jenoobi, F.I. Delivery of Insulin via Skin Route for the Management of Diabetes Mellitus: Approaches for Breaching the Obstacles. Pharmaceutics 2021, 13, 100. [Google Scholar] [CrossRef]
- Jeong, S.-Y.; Park, J.-H.; Lee, Y.-S.; Kim, Y.-S.; Park, J.-Y.; Kim, S.-Y. The Current Status of Clinical Research Involving Microneedles: A Systematic Review. Pharmaceutics 2020, 12, 1113. [Google Scholar] [CrossRef]
- Oleck, J.; Kassam, S.; Goldman, J.D. Commentary: Why Was Inhaled Insulin a Failure in the Market? Diabetes Spectr. 2016, 29, 180–184. [Google Scholar] [CrossRef] [Green Version]
- Lipska, K.J.; Ross, J.S.; Van Houten, H.K.; Beran, D.; Yudkin, J.S.; Shah, N.D. Use and Out-of-Pocket Costs of Insulin for Type 2 Diabetes Mellitus From 2000 Through 2010. JAMA 2014, 311, 2331. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Zhao, J.J.; Mitlak, B.H.; Wang, O.; Genant, H.K.; Eriksen, E.F. Recombinant Human Parathyroid Hormone (1–34) [Teriparatide] Improves Both Cortical and Cancellous Bone Structure. J. Bone Miner. Res. 2003, 18, 1932–1941. [Google Scholar] [CrossRef] [PubMed]
- Reeve, J. Recombinant human parathyroid hormone. BMJ 2002, 324, 435–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLaughlin, M.B.; Jialal, I. Calcitonin. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Sun, L.-M.; Lin, M.-C.; Muo, C.-H.; Liang, J.-A.; Kao, C.-H. Calcitonin Nasal Spray and Increased Cancer Risk: A Population-Based Nested Case-Control Study. J. Clin. Endocrinol. Metab. 2014, 99, 4259–4264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, M.; Toplak, A.; Quaedflieg, P.J.; Nuijens, T. Enzyme-mediated ligation technologies for peptides and proteins. Curr. Opin. Chem. Biol. 2017, 38, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Iype, M.; Koshy, K.G. Management of Infantile Spasms: An Updated Review. Int. J. Epilepsy 2020, 6, 4–14. [Google Scholar] [CrossRef]
- Anderson, S.L.; Beutel, T.R.; Trujillo, J.M. Oral semaglutide in type 2 diabetes. J. Diabetes Complicat. 2020, 34, 107520. [Google Scholar] [CrossRef]
- Knudsen, L.B.; Lau, J. The Discovery and Development of Liraglutide and Semaglutide. Front. Endocrinol. 2019, 10, 155. [Google Scholar] [CrossRef] [Green Version]
- Labadzhyan, A.; Nachtigall, L.B.; Fleseriu, M.; Gordon, M.B.; Molitch, M.; Kennedy, L.; Samson, S.L.; Greenman, Y.; Biermasz, N.; Bolanowski, M.; et al. Correction to: Oral octreotide capsules for the treatment of acromegaly: Comparison of 2 phase 3 trial results. Pituitary 2021, 24, 943–953. [Google Scholar] [CrossRef]
- Mullard, A. 2021 FDA approvals. Nat. Rev. Drug Discov. 2022. [Google Scholar] [CrossRef]
- Russo, A.F. Overview of Neuropeptides: Awakening the Senses? Headache J. Head Face Pain 2017, 57, 37–46. [Google Scholar] [CrossRef]
- Fricker, L.D.; Margolis, E.B.; Gomes, I.; Devi, L.A. Five Decades of Research on Opioid Peptides: Current Knowledge and Unanswered Questions. Mol. Pharmacol. 2020, 98, 96–108. [Google Scholar] [CrossRef]
- Merighi, A.; Salio, C.; Ferrini, F.; Lossi, L. Neuromodulatory function of neuropeptides in the normal CNS. J. Chem. Neuroanat. 2011, 42, 276–287. [Google Scholar] [CrossRef]
- Barg, S.; Olofsson, C.S.; Schriever-Abeln, J.; Wendt, A.; Gebre-Medhin, S.; Renström, E.; Rorsman, P. Delay between Fusion Pore Opening and Peptide Release from Large Dense-Core Vesicles in Neuroendocrine Cells. Neuron 2002, 33, 287–299. [Google Scholar] [CrossRef] [Green Version]
- Catalani, E.; De Palma, C.; Perrotta, C.; Cervia, D. Current Evidence for a Role of Neuropeptides in the Regulation of Autophagy. BioMed Res. Int. 2017, 2017, 5856071. [Google Scholar] [CrossRef]
- Guillaumin, M.C.C.; Burdakov, D. Neuropeptides as Primary Mediators of Brain Circuit Connectivity. Front. Neurosci. 2021, 15, 644313. [Google Scholar] [CrossRef] [PubMed]
- Souza-Moreira, L.; Campos-Salinas, J.; Caro, M.; Gonzalez-Rey, E. Neuropeptides as Pleiotropic Modulators of the Immune Response. Neuroendocrinology 2011, 94, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Davenport, A.P.; Scully, C.C.G.; de Graaf, C.; Brown, A.J.H.; Maguire, J.J. Advances in therapeutic peptides targeting G protein-coupled receptors. Nat. Rev. Drug Discov. 2020, 19, 389–413. [Google Scholar] [CrossRef] [PubMed]
- Valenstein, E.S. The Discovery of Chemical Neurotransmitters. Brain Cogn. 2002, 49, 73–95. [Google Scholar] [CrossRef]
- Krehbiel, D.; Bartel, B.; Dirks, M.; Wiens, W. Behavior and brain neurotransmitters: Correlations in different strains of mice. Behav. Neural Biol. 1986, 46, 30–45. [Google Scholar] [CrossRef]
- Hanin, I. Central neurotransmitter function and its behavioral correlates in man. Environ. Health Perspect. 1978, 26, 135–141. [Google Scholar] [CrossRef]
- Aghajanian, G. Serotonin and Hallucinogens. Neuropsychopharmacology 1999, 21, 16–23. [Google Scholar] [CrossRef] [Green Version]
- Coyle, J.; Kershaw, P. Galantamine, a cholinesterase inhibitor that allosterically modulates nicotinic receptors: Effects on the course of Alzheimer’s disease. Biol. Psychiatry 2001, 49, 289–299. [Google Scholar] [CrossRef]
- Byerley, W.F.; Reimherr, F.W.; Wood, D.R.; Grosser, B.I. Fluoxetine, a selective serotonin uptake inhibitor, for the treatment of outpatients with major depression. J. Clin. Psychopharmacol. 1988, 8, 112–115. [Google Scholar] [CrossRef]
- Hill, J. Vasoactive Intestinal Peptide in Neurodevelopmental Disorders:Therapeutic Potential. Curr. Pharm. Des. 2007, 13, 1079–1089. [Google Scholar] [CrossRef] [Green Version]
- Passemard, S.; El Ghouzzi, V.; Nasser, H.; Verney, C.; Vodjdani, G.; Lacaud, A.; Lebon, S.; Laburthe, M.; Robberecht, P.; Nardelli, J.; et al. VIP blockade leads to microcephaly in mice via disruption of Mcph1-Chk1 signaling. J. Clin. Investig. 2011, 121, 3072–3087. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.Y.; Henins, K.A.; Gressens, P.; Gozes, I.; Fridkin, M.; Brenneman, D.E.; Hill, J.M. Neurobehavioral Development of Neonatal Mice Following Blockade of VIP During the Early Embryonic Period. Peptides 1997, 18, 1131–1137. [Google Scholar] [CrossRef]
- Nelson, K.B.; Grether, J.K.; Croen, L.A.; Dambrosia, J.M.; Dickens, B.F.; Jelliffe, L.L.; Hansen, R.L.; Phillips, T.M. Neuropeptides and neurotrophins in neonatal blood of children with autism or mental retardation. Ann. Neurol. 2001, 49, 597–606. [Google Scholar] [CrossRef]
- Levinson, D.F.; Duan, J.; Oh, S.; Wang, K.; Sanders, A.R.; Shi, J.; Zhang, N.; Mowry, B.J.; Olincy, A.; Amin, F.; et al. Copy Number Variants in Schizophrenia: Confirmation of Five Previous Findings and New Evidence for 3q29 Microdeletions and VIPR2 Duplications. Am. J. Psychiatry 2011, 168, 302–316. [Google Scholar] [CrossRef] [Green Version]
- Firouzabadi, S.G.; Kariminejad, R.; Vameghi, R.; Darvish, H.; Ghaedi, H.; Banihashemi, S.; Firouzkouhi Moghaddam, M.; Jamali, P.; Mofidi Tehrani, H.F.; Dehghani, H.; et al. Copy Number Variants in Patients with Autism and Additional Clinical Features: Report of VIPR2 Duplication and a Novel Microduplication Syndrome. Mol. Neurobiol. 2017, 54, 7019–7027. [Google Scholar] [CrossRef]
- Hill, J.M.; McCune, S.K.; Alvero, R.J.; Glazner, G.W.; Henins, K.A.; Stanziale, S.F.; Keimowitz, J.R.; Brenneman, D.E. Maternal vasoactive intestinal peptide and the regulation of embryonic growth in the rodent. J. Clin. Investig. 1996, 97, 202–208. [Google Scholar] [CrossRef] [Green Version]
- Lorsung, E.; Karthikeyan, R.; Cao, R. Biological Timing and Neurodevelopmental Disorders: A Role for Circadian Dysfunction in Autism Spectrum Disorders. Front. Neurosci. 2021, 15, 642745. [Google Scholar] [CrossRef]
- Ono, D.; Honma, K.; Honma, S. Roles of Neuropeptides, VIP and AVP, in the Mammalian Central Circadian Clock. Front. Neurosci. 2021, 15, 351. [Google Scholar] [CrossRef]
- Mazuski, C.; Chen, S.P.; Herzog, E.D. Different Roles for VIP Neurons in the Neonatal and Adult Suprachiasmatic Nucleus. J. Biol. Rhythms 2020, 35, 465–475. [Google Scholar] [CrossRef]
- Muscatelli, F.; Desarménien, M.G.; Matarazzo, V.; Grinevich, V. Oxytocin Signaling in the Early Life of Mammals: Link to Neurodevelopmental Disorders Associated with ASD. In Behavioral Pharmacology of Neuropeptides: Oxytocin; Hurlemann, R., Grinevich, V., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 239–268. ISBN 978-3-319-63739-6. [Google Scholar]
- Miller, T.V.; Caldwell, H.K. Oxytocin during Development: Possible Organizational Effects on Behavior. Front. Endocrinol. 2015, 6, 76. [Google Scholar] [CrossRef] [Green Version]
- LoParo, D.; Waldman, I.D. The oxytocin receptor gene (OXTR) is associated with autism spectrum disorder: A meta-analysis. Mol. Psychiatry 2015, 20, 640–646. [Google Scholar] [CrossRef]
- Lerer, E.; Levi, S.; Salomon, S.; Darvasi, A.; Yirmiya, N.; Ebstein, R.P. Association between the oxytocin receptor (OXTR) gene and autism: Relationship to Vineland Adaptive Behavior Scales and cognition. Mol. Psychiatry 2008, 13, 980–988. [Google Scholar] [CrossRef] [Green Version]
- Guastella, A.J.; Hickie, I.B. Oxytocin Treatment, Circuitry, and Autism: A Critical Review of the Literature Placing Oxytocin Into the Autism Context. Biol. Psychiatry 2016, 79, 234–242. [Google Scholar] [CrossRef]
- Rajamani, K.T.; Wagner, S.; Grinevich, V.; Harony-Nicolas, H. Oxytocin as a Modulator of Synaptic Plasticity: Implications for Neurodevelopmental Disorders. Front. Synaptic Neurosci. 2018, 10, 17. [Google Scholar] [CrossRef] [Green Version]
- Sahay, A.; Kale, A.; Joshi, S. Role of neurotrophins in pregnancy and offspring brain development. Neuropeptides 2020, 83, 102075. [Google Scholar] [CrossRef]
- Maduna, T.; Lelievre, V. Neuropeptides shaping the central nervous system development: Spatiotemporal actions of VIP and PACAP through complementary signaling pathways: Neuropeptides Shaping Up CNS Development. J. Neurosci. Res. 2016, 94, 1472–1487. [Google Scholar] [CrossRef]
- Thakker-Varia, S.; Behnke, J.; Doobin, D.; Dalal, V.; Thakkar, K.; Khadim, F.; Wilson, E.; Palmieri, A.; Antila, H.; Rantamaki, T.; et al. VGF (TLQP-62)-induced neurogenesis targets early phase neural progenitor cells in the adult hippocampus and requires glutamate and BDNF signaling. Stem Cell Res. 2014, 12, 762–777. [Google Scholar] [CrossRef] [Green Version]
- Jinno, S.; Kosaka, T. Patterns of expression of neuropeptides in GABAergic nonprincipal neurons in the mouse hippocampus: Quantitative analysis with optical disector. J. Comp. Neurol. 2003, 461, 333–349. [Google Scholar] [CrossRef]
- Xu, X.; Guo, F.; He, Q.; Cai, X.; Min, D.; Wang, Q.; Wang, S.; Tian, L.; Cai, J.; Zhao, Y. Altered expression of neuropeptide Y, Y1 and Y2 receptors, but not Y5 receptor, within hippocampus and temporal lobe cortex of tremor rats. Neuropeptides 2014, 48, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Howell, O.W.; Silva, S.; Scharfman, H.E.; Sosunov, A.A.; Zaben, M.; Shatya, A.; Mckhann, G.; Herzog, H.; Laskowski, A.; Gray, W.P. Neuropeptide Y is important for basal and seizure-induced precursor cell proliferation in the hippocampus. Neurobiol. Dis. 2007, 26, 174–188. [Google Scholar] [CrossRef] [PubMed]
- Howell, O.W.; Doyle, K.; Goodman, J.H.; Scharfman, H.E.; Herzog, H.; Pringle, A.; Beck-Sickinger, A.G.; Gray, W.P. Neuropeptide Y stimulates neuronal precursor proliferation in the post-natal and adult dentate gyrus. J. Neurochem. 2005, 93, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Hansel, D.E.; Eipper, B.A.; Ronnett, G.V. Neuropeptide Y functions as a neuroproliferative factor. Nature 2001, 410, 940–944. [Google Scholar] [CrossRef] [PubMed]
- Geloso, M.C.; Corvino, V.; Di Maria, V.; Marchese, E.; Michetti, F. Cellular targets for neuropeptide Y-mediated control of adult neurogenesis. Front. Cell. Neurosci. 2015, 9, 85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karl, T.; Duffy, L.; Herzog, H. Behavioural profile of a new mouse model for NPY deficiency: Behavioural profile of a new mouse for NPY deficiency. Eur. J. Neurosci. 2008, 28, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Sun, G.; Zhao, C.; Stewart, R. Neural stem cell self-renewal. Crit. Rev. Oncol. Hematol. 2008, 65, 43–53. [Google Scholar] [CrossRef] [Green Version]
- Furtinger, S.; Pirker, S.; Czech, T.; Baumgartner, C.; Ransmayr, G.; Sperk, G. Plasticity of Y1 and Y2 Receptors and Neuropeptide Y Fibers in Patients with Temporal Lobe Epilepsy. J. Neurosci. 2001, 21, 5804–5812. [Google Scholar] [CrossRef] [Green Version]
- Yin, J.; Yuan, Q. Structural homeostasis in the nervous system: A balancing act for wiring plasticity and stability. Front. Cell. Neurosci. 2015, 8, 439. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Wang, M.; Yin, S.; Jang, R.; Wang, J.; Xue, Z.; Xu, T. NeuroPep: A comprehensive resource of neuropeptides. Database 2015, 2015, bav038. [Google Scholar] [CrossRef] [Green Version]
- Burbach, J.P.H. Neuropeptides from concept to online database www.neuropeptides.nl. Eur. J. Pharmacol. 2010, 626, 27–48. [Google Scholar] [CrossRef]
- Scheller, R.H.; Jackson, J.F.; McAllister, L.B.; Rothman, B.S.; Mayeri, E.; Axel, R. A single gene encodes multiple neuropeptides mediating a stereotyped behavior. Cell 1983, 32, 7–22. [Google Scholar] [CrossRef]
- Zhang, X. Neuropeptide Regulation of Ion Channels and Food Intake. In Nonclassical Ion Channels in the Nervous System; CRC Press: Boca Raton, FL, USA, 2021; ISBN 978-1-00-310926-6. [Google Scholar]
- DeLaney, K.; Buchberger, A.R.; Atkinson, L.; Gründer, S.; Mousley, A.; Li, L. New techniques, applications and perspectives in neuropeptide research. J. Exp. Biol. 2018, 221, jeb151167. [Google Scholar] [CrossRef] [Green Version]
- Cottrell, G.A.; Green, K.A.; Davies, N.W. The neuropeptide Phe-Met-Arg-Phe-NH2 (FMRFamide) can activate a ligand-gated ion channel in Helix neurons. Pflügers Arch. 1990, 416, 612–614. [Google Scholar] [CrossRef]
- Osmakov, D.I.; Koshelev, S.G.; Ivanov, I.A.; Andreev, Y.A.; Kozlov, S.A. Endogenous Neuropeptide Nocistatin Is a Direct Agonist of Acid-Sensing Ion Channels (ASIC1, ASIC2 and ASIC3). Biomolecules 2019, 9, 401. [Google Scholar] [CrossRef] [Green Version]
- Kuspiel, S.; Wiemuth, D.; Gründer, S. The Neuropeptide Nocistatin Is Not a Direct Agonist of Acid-Sensing Ion Channel 1a (ASIC1a). Biomolecules 2021, 11, 571. [Google Scholar] [CrossRef]
- Leisle, L.; Margreiter, M.; Ortega-Ramírez, A.; Cleuvers, E.; Bachmann, M.; Rossetti, G.; Gründer, S. Dynorphin Neuropeptides Decrease Apparent Proton Affinity of ASIC1a by Occluding the Acidic Pocket. J. Med. Chem. 2021, 64, 13299–13311. [Google Scholar] [CrossRef]
- Reiners, M.; Margreiter, M.A.; Oslender-Bujotzek, A.; Rossetti, G.; Gründer, S.; Schmidt, A. The Conorfamide RPRFa Stabilizes the Open Conformation of Acid-Sensing Ion Channel 3 via the Nonproton Ligand–Sensing Domain. Mol. Pharmacol. 2018, 94, 1114–1124. [Google Scholar] [CrossRef] [Green Version]
- Xiong, Z.-G.; Zhu, X.-M.; Chu, X.-P.; Minami, M.; Hey, J.; Wei, W.-L.; MacDonald, J.F.; Wemmie, J.A.; Price, M.P.; Welsh, M.J.; et al. Neuroprotection in Ischemia. Cell 2004, 118, 687–698. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; You, Z.; Wang, S.; Yang, J.; Yang, L.; Sun, Y.; Mi, W.; Yang, L.; McCabe, M.F.; Shen, S.; et al. Neuropeptide S modulates the amygdaloidal HCN activities (Ih) in rats: Implication in chronic pain. Neuropharmacology 2016, 105, 420–433. [Google Scholar] [CrossRef]
- Simmons, M.L.; Terman, G.W.; Gibbs, S.M.; Chavkin, C. L-type calcium channels mediate dynorphin neuropeptide release from dendrites but not axons of hippocampal granule cells. Neuron 1995, 14, 1265–1272. [Google Scholar] [CrossRef] [Green Version]
- Wagner, J.; Caudle, R.; Chavkin, C. Kappa-opioids decrease excitatory transmission in the dentate gyrus of the guinea pig hippocampus. J. Neurosci. 1992, 12, 132–141. [Google Scholar] [CrossRef] [Green Version]
- Iremonger, K.J.; Bains, J.S. Retrograde Opioid Signaling Regulates Glutamatergic Transmission in the Hypothalamus. J. Neurosci. 2009, 29, 7349–7358. [Google Scholar] [CrossRef] [Green Version]
- Busciglio, J.; Gabuzda, D.H.; Matsudaira, P.; Yankner, B.A. Generation of beta-amyloid in the secretory pathway in neuronal and nonneuronal cells. Proc. Natl. Acad. Sci. USA 1993, 90, 2092–2096. [Google Scholar] [CrossRef] [Green Version]
- Toneff, T.; Funkelstein, L.; Mosier, C.; Abagyan, A.; Ziegler, M.; Hook, V. Beta-amyloid peptides undergo regulated co-secretion with neuropeptide and catecholamine neurotransmitters. Peptides 2013, 46, 126–135. [Google Scholar] [CrossRef] [Green Version]
- Puzzo, D.; Privitera, L.; Leznik, E.; Fa, M.; Staniszewski, A.; Palmeri, A.; Arancio, O. Picomolar Amyloid- Positively Modulates Synaptic Plasticity and Memory in Hippocampus. J. Neurosci. 2008, 28, 14537–14545. [Google Scholar] [CrossRef]
- Finnie, P.S.B.; Nader, K. Amyloid Beta Secreted during Consolidation Prevents Memory Malleability. Curr. Biol. 2020, 30, 1934–1940.e4. [Google Scholar] [CrossRef]
- Kuzuya, A.; Zoltowska, K.M.; Post, K.L.; Arimon, M.; Li, X.; Svirsky, S.; Maesako, M.; Muzikansky, A.; Gautam, V.; Kovacs, D.; et al. Identification of the novel activity-driven interaction between synaptotagmin 1 and presenilin 1 links calcium, synapse, and amyloid beta. BMC Biol. 2016, 14, 25. [Google Scholar] [CrossRef] [Green Version]
- Parihar, M.S.; Brewer, G.J. Amyloid-β as a Modulator of Synaptic Plasticity. J. Alzheimers Dis. 2010, 22, 741–763. [Google Scholar] [CrossRef] [Green Version]
- Puzzo, D.; Gulisano, W.; Arancio, O.; Palmeri, A. The keystone of Alzheimer pathogenesis might be sought in Aβ physiology. Neuroscience 2015, 307, 26–36. [Google Scholar] [CrossRef] [Green Version]
- Alder, J.; Kallman, S.; Palmieri, A.; Khadim, F.; Ayer, J.J.; Kumar, S.; Tsung, K.; Grinberg, I.; Thakker-Varia, S. Neuropeptide orphanin FQ inhibits dendritic morphogenesis through activation of RhoA: OFQ Inhibits Dendrite Growth via RhoA. Dev. Neurobiol. 2013, 73, 769–784. [Google Scholar] [CrossRef]
- Seseña, E.; Soto, E.; Bueno, J.; Vega, R. Nociceptin/orphanin FQ peptide receptor mediates inhibition of N-type calcium currents in vestibular afferent neurons of the rat. J. Neurophysiol. 2020, 124, 1605–1614. [Google Scholar] [CrossRef]
- Frank, C.A. How voltage-gated calcium channels gate forms of homeostatic synaptic plasticity. Front. Cell. Neurosci. 2014, 8, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Fruyt, N.; Yu, A.J.; Rankin, C.H.; Beets, I.; Chew, Y.L. The role of neuropeptides in learning: Insights from C. elegans. Int. J. Biochem. Cell Biol. 2020, 125, 105801. [Google Scholar] [CrossRef] [PubMed]
- Peineau, S.; Rabiant, K.; Pierrefiche, O.; Potier, B. Synaptic plasticity modulation by circulating peptides and metaplasticity: Involvement in Alzheimer’s disease. Pharmacol. Res. 2018, 130, 385–401. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, A.; Letzkus, J.J. A neuropeptide making memories. Cell 2021, 184, 5501–5503. [Google Scholar] [CrossRef]
- Kash, T.L.; Winder, D.G. Neuropeptide Y and corticotropin-releasing factor bi-directionally modulate inhibitory synaptic transmission in the bed nucleus of the stria terminalis. Neuropharmacology 2006, 51, 1013–1022. [Google Scholar] [CrossRef]
- Wood, J.; Verma, D.; Lach, G.; Bonaventure, P.; Herzog, H.; Sperk, G.; Tasan, R.O. Structure and function of the amygdaloid NPY system: NPY Y2 receptors regulate excitatory and inhibitory synaptic transmission in the centromedial amygdala. Brain Struct. Funct. 2016, 221, 3373–3391. [Google Scholar] [CrossRef] [Green Version]
- Pomrenze, M.B.; Giovanetti, S.M.; Maiya, R.; Gordon, A.G.; Kreeger, L.J.; Messing, R.O. Dissecting the Roles of GABA and Neuropeptides from Rat Central Amygdala CRF Neurons in Anxiety and Fear Learning. Cell Rep. 2019, 29, 13–21.e4. [Google Scholar] [CrossRef] [Green Version]
- Bartsch, T.; Schonfeld, R.; Muller, F.J.; Alfke, K.; Leplow, B.; Aldenhoff, J.; Deuschl, G.; Koch, J.M. Focal Lesions of Human Hippocampal CA1 Neurons in Transient Global Amnesia Impair Place Memory. Science 2010, 328, 1412–1415. [Google Scholar] [CrossRef]
- Wang, X.F.; Ge, T.T.; Fan, J.; Yang, W.; Li, B.; Cui, R.J. The role of substance P in epilepsy and seizure disorders. Oncotarget 2017, 8, 78225–78233. [Google Scholar] [CrossRef] [Green Version]
- Menon, N.; Prabhavalkar, K.S.; Bhatt, L.K. Neuropeptides: A promising target for treating seizures. Neuropeptides 2017, 65, 63–70. [Google Scholar] [CrossRef]
- Neugebauer, V. Amygdala pain mechanisms. Handb. Exp. Pharmacol. 2015, 227, 261–284. [Google Scholar] [CrossRef] [Green Version]
- Neugebauer, V.; Mazzitelli, M.; Cragg, B.; Ji, G.; Navratilova, E.; Porreca, F. Amygdala, neuropeptides, and chronic pain-related affective behaviors. Neuropharmacology 2020, 170, 108052. [Google Scholar] [CrossRef]
- Ikeda, R.; Takahashi, Y.; Inoue, K.; Kato, F. NMDA receptor-independent synaptic plasticity in the central amygdala in the rat model of neuropathic pain. Pain 2007, 127, 161–172. [Google Scholar] [CrossRef]
- Sugimura, Y.K.; Takahashi, Y.; Watabe, A.M.; Kato, F. Synaptic and network consequences of monosynaptic nociceptive inputs of parabrachial nucleus origin in the central amygdala. J. Neurophysiol. 2016, 115, 2721–2739. [Google Scholar] [CrossRef] [Green Version]
- Shinohara, K.; Watabe, A.M.; Nagase, M.; Okutsu, Y.; Takahashi, Y.; Kurihara, H.; Kato, F. Essential role of endogenous calcitonin gene-related peptide in pain-associated plasticity in the central amygdala. Eur. J. Neurosci. 2017, 46, 2149–2160. [Google Scholar] [CrossRef] [Green Version]
- Han, J.S.; Adwanikar, H.; Li, Z.; Ji, G.; Neugebauer, V. Facilitation of Synaptic Transmission and Pain Responses by CGRP in the Amygdala of Normal Rats. Mol. Pain 2010, 6, 1744–8069. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, Y.-L.; Wu, B.-T.; Yang, C.-C. Increased substance P-like immunoreactivities in parabrachial and amygdaloid nuclei in a rat model with masticatory myofascial pain. Exp. Brain Res. 2020, 238, 2845–2855. [Google Scholar] [CrossRef]
- Stoop, R.; Hegoburu, C.; van den Burg, E. New Opportunities in Vasopressin and Oxytocin Research: A Perspective from the Amygdala. Annu. Rev. Neurosci. 2015, 38, 369–388. [Google Scholar] [CrossRef]
- Cragg, B.; Ji, G.; Neugebauer, V. Differential contributions of vasopressin V1A and oxytocin receptors in the amygdala to pain-related behaviors in rats. Mol. Pain 2016, 12, 174480691667649. [Google Scholar] [CrossRef] [Green Version]
- Boyle, C.A.; Hu, B.; Quaintance, K.L.; Lei, S. Involvement of TRPC5 channels, inwardly rectifying K+ channels, PLCβ and PIP2 in vasopressin-mediated excitation of medial central amygdala neurons. J. Physiol. 2021, 599, 3101–3119. [Google Scholar] [CrossRef]
- Wilson, T.D.; Valdivia, S.; Khan, A.; Ahn, H.-S.; Adke, A.P.; Martinez Gonzalez, S.; Sugimura, Y.K.; Carrasquillo, Y. Dual and Opposing Functions of the Central Amygdala in the Modulation of Pain. Cell Rep. 2019, 29, 332–346.e5. [Google Scholar] [CrossRef] [Green Version]
- Medina, G.; Ji, G.; Grégoire, S.; Neugebauer, V. Nasal Application of Neuropeptide S Inhibits Arthritis Pain-Related Behaviors through an Action in the Amygdala. Mol. Pain 2014, 10, 1744–8069. [Google Scholar] [CrossRef] [Green Version]
- Ji, G.; Neugebauer, V. Pro- and Anti-Nociceptive Effects of Corticotropin-Releasing Factor (CRF) in Central Amygdala Neurons Are Mediated Through Different Receptors. J. Neurophysiol. 2008, 99, 1201–1212. [Google Scholar] [CrossRef] [Green Version]
- Sprouse-Blum, A.S.; Smith, G.; Sugai, D.; Parsa, F.D. Understanding endorphins and their importance in pain management. Hawaii Med. J. 2010, 69, 70–71. [Google Scholar]
- Carroll, R.G. 13-Endocrine System. In Elsevier’s Integrated Physiology; Carroll, R.G., Ed.; Mosby: Philadelphia, PA, USA, 2007; pp. 157–176. ISBN 978-0-323-04318-2. [Google Scholar]
- Struthers, A.D.; Brown, M.J.; Macdonald, D.W.R.; Beacham, J.L.; Stevenson, J.C.; Morris, H.R.; Macintyre, I. Human calcitonin gene related peptide: A potent endogenous vasodilator in man. Clin. Sci. 1986, 70, 389–393. [Google Scholar] [CrossRef] [Green Version]
- Brain, S.D.; Williams, T.J.; Tippins, J.R.; Morris, H.R.; MacIntyre, I. Calcitonin gene-related peptide is a potent vasodilator. Nature 1985, 313, 54–56. [Google Scholar] [CrossRef]
- Lembeck, F.; Holzer, P. Substance P as neurogenic mediator of antidromic vasodilation and neurogenic plasma extravasation. Naunyn. Schmiedebergs Arch. Pharmacol. 1979, 310, 175–183. [Google Scholar] [CrossRef]
- Kee, Z.; Kodji, X.; Brain, S.D. The Role of Calcitonin Gene Related Peptide (CGRP) in Neurogenic Vasodilation and Its Cardioprotective Effects. Front. Physiol. 2018, 9, 1249. [Google Scholar] [CrossRef] [Green Version]
- Czerwińska, M.; Czarzasta, K.; Cudnoch-Jędrzejewska, A. New Peptides as Potential Players in the Crosstalk Between the Brain and Obesity, Metabolic and Cardiovascular Diseases. Front. Physiol. 2021, 12, 692642. [Google Scholar] [CrossRef] [PubMed]
- Carr, R.; Frings, S. Neuropeptides in sensory signal processing. Cell Tissue Res. 2019, 375, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Genovese, F.; Bauersachs, H.G.; Gräßer, I.; Kupke, J.; Magin, L.; Daiber, P.; Möhrlen, F.; Messlinger, K.; Frings, S. Possible role of calcitonin gene-related peptide in trigeminal modulation of glomerular microcircuits of the rodent olfactory bulb. Eur. J. Neurosci. 2017, 45, 587–600. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, M.; Imura, K.; Sato, I. Topographical organization of TRPV1-immunoreactive epithelium and CGRP-immunoreactive nerve terminals in rodent tongue. Eur. J. Histochem. EJH 2012, 56, e21. [Google Scholar] [CrossRef] [PubMed]
- Blixt, F.W.; Radziwon-Balicka, A.; Edvinsson, L.; Warfvinge, K. Distribution of CGRP and its receptor components CLR and RAMP1 in the rat retina. Exp. Eye Res. 2017, 161, 124–131. [Google Scholar] [CrossRef]
- Dickerson, I.M.; Bussey-Gaborski, R.; Holt, J.C.; Jordan, P.M.; Luebke, A.E. Maturation of suprathreshold auditory nerve activity involves cochlear CGRP-receptor complex formation. Physiol. Rep. 2016, 4, e12869. [Google Scholar] [CrossRef] [Green Version]
- Riera, C.E.; Dillin, A. Emerging Role of Sensory Perception in Aging and Metabolism. Trends Endocrinol. Metab. 2016, 27, 294–303. [Google Scholar] [CrossRef]
- Stevenson, R.A.; Park, S.; Cochran, C.; McIntosh, L.G.; Noel, J.-P.; Barense, M.D.; Ferber, S.; Wallace, M.T. The associations between multisensory temporal processing and symptoms of schizophrenia. Schizophr. Res. 2017, 179, 97–103. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.; Petersen, M.; Hoyer, N.; Spitzweck, B.; Tenedini, F.; Wang, D.; Gruschka, A.; Burchardt, L.S.; Szpotowicz, E.; Schweizer, M.; et al. Sensory integration and neuromodulatory feedback facilitate Drosophila mechanonociceptive behavior. Nat. Neurosci. 2017, 20, 1085–1095. [Google Scholar] [CrossRef] [Green Version]
- Graham, C.E.; Basappa, J.; Vetter, D.E. A corticotropin-releasing factor system expressed in the cochlea modulates hearing sensitivity and protects against noise-induced hearing loss. Neurobiol. Dis. 2010, 38, 246–258. [Google Scholar] [CrossRef] [Green Version]
- Pagella, S.; Deussing, J.M.; Kopp-Scheinpflug, C. Expression Patterns of the Neuropeptide Urocortin 3 and Its Receptor CRFR2 in the Mouse Central Auditory System. Front. Neural Circuits 2021, 15, 747472. [Google Scholar] [CrossRef]
- Carniglia, L.; Ramírez, D.; Durand, D.; Saba, J.; Turati, J.; Caruso, C.; Scimonelli, T.N.; Lasaga, M. Neuropeptides and Microglial Activation in Inflammation, Pain, and Neurodegenerative Diseases. Mediat. Inflamm. 2017, 2017, 5048616. [Google Scholar] [CrossRef]
- Xu, H.; Shi, X.; Li, X.; Zou, J.; Zhou, C.; Liu, W.; Shao, H.; Chen, H.; Shi, L. Neurotransmitter and neuropeptide regulation of mast cell function: A systematic review. J. Neuroinflammation 2020, 17, 356. [Google Scholar] [CrossRef]
- Cocchiara, R.; Lampiasi, N.; Albeggiani, G.; Bongiovanni, A.; Azzolina, A.; Geraci, D. Mast cell production of TNF-α induced by substance P evidence for a modulatory role of substance P-antagonists. J. Neuroimmunol. 1999, 101, 128–136. [Google Scholar] [CrossRef]
- Azzolina, A.; Bongiovanni, A.; Lampiasi, N. Substance P induces TNF-α and IL-6 production through NFκB in peritoneal mast cells. Biochim. Biophys. Acta BBA Mol. Cell Res. 2003, 1643, 75–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Voirin, A.-C.; Perek, N.; Roche, F. Inflammatory stress induced by a combination of cytokines (IL-6, IL-17, TNF-α) leads to a loss of integrity on bEnd.3 endothelial cells in vitro BBB model. Brain Res. 2020, 1730, 146647. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, H.; Gupta, K.; Ali, H. Roles of Mas-related G protein–coupled receptor X2 on mast cell–mediated host defense, pseudoallergic drug reactions, and chronic inflammatory diseases. J. Allergy Clin. Immunol. 2016, 138, 700–710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cunningham, C.; Campion, S.; Lunnon, K.; Murray, C.L.; Woods, J.F.C.; Deacon, R.M.J.; Rawlins, J.N.P.; Perry, V.H. Systemic Inflammation Induces Acute Behavioral and Cognitive Changes and Accelerates Neurodegenerative Disease. Biol. Psychiatry 2009, 65, 304–312. [Google Scholar] [CrossRef] [Green Version]
- Giridharan, V.V.; Masud, F.; Petronilho, F.; Dal-Pizzol, F.; Barichello, T. Infection-Induced Systemic Inflammation Is a Potential Driver of Alzheimer’s Disease Progression. Front. Aging Neurosci. 2019, 11, 122. [Google Scholar] [CrossRef]
- Walker, K.A.; Ficek, B.N.; Westbrook, R. Understanding the Role of Systemic Inflammation in Alzheimer’s Disease. ACS Chem. Neurosci. 2019, 10, 3340–3342. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.L.; Rodriguez-Ortiz, C.J.; Kitazawa, M. Infection, systemic inflammation, and Alzheimer’s disease. Microbes Infect. 2015, 17, 549–556. [Google Scholar] [CrossRef]
- Dziedzic, T. Systemic inflammation as a therapeutic target in acute ischemic stroke. Expert Rev. Neurother. 2015, 15, 523–531. [Google Scholar] [CrossRef]
- Esenwa, C.C.; Elkind, M.S. Inflammatory risk factors, biomarkers and associated therapy in ischaemic stroke. Nat. Rev. Neurol. 2016, 12, 594–604. [Google Scholar] [CrossRef]
- Adams, B.; Nunes, J.M.; Page, M.J.; Roberts, T.; Carr, J.; Nell, T.A.; Kell, D.B.; Pretorius, E. Parkinson’s Disease: A Systemic Inflammatory Disease Accompanied by Bacterial Inflammagens. Front. Aging Neurosci. 2019, 11, 210. [Google Scholar] [CrossRef] [Green Version]
- Fan, Z.; Pan, Y.-T.; Zhang, Z.-Y.; Yang, H.; Yu, S.-Y.; Zheng, Y.; Ma, J.-H.; Wang, X.-M. Systemic activation of NLRP3 inflammasome and plasma α-synuclein levels are correlated with motor severity and progression in Parkinson’s disease. J. Neuroinflammation 2020, 17, 11. [Google Scholar] [CrossRef] [Green Version]
- McCauley, M.E.; Baloh, R.H. Inflammation in ALS/FTD pathogenesis. Acta Neuropathol. 2019, 137, 715–730. [Google Scholar] [CrossRef] [Green Version]
- Valadão, P.A.C.; Santos, K.B.S.; Ferreira e Vieira, T.H.; Macedo e Cordeiro, T.; Teixeira, A.L.; Guatimosim, C.; de Miranda, A.S. Inflammation in Huntington’s disease: A few new twists on an old tale. J. Neuroimmunol. 2020, 348, 577380. [Google Scholar] [CrossRef]
- Sun, C.; An, Q.; Li, R.; Chen, S.; Gu, X.; An, S.; Wang, Z. Calcitonin gene-related peptide induces the histone H3 lysine 9 acetylation in astrocytes associated with neuroinflammation in rats with neuropathic pain. CNS Neurosci. Ther. 2021, 27, 1409–1424. [Google Scholar] [CrossRef]
- Escobedo, G.; López-Ortiz, E.; Torres-Castro, I. Gut microbiota as a key player in triggering obesity, systemic inflammation and insulin resistance. Rev. Investig. Clin. Organo Hosp. Enfermedades Nutr. 2014, 66, 450–459. [Google Scholar]
- Cussotto, S.; Delgado, I.; Anesi, A.; Dexpert, S.; Aubert, A.; Beau, C.; Forestier, D.; Ledaguenel, P.; Magne, E.; Mattivi, F.; et al. Tryptophan Metabolic Pathways Are Altered in Obesity and Are Associated With Systemic Inflammation. Front. Immunol. 2020, 11, 557. [Google Scholar] [CrossRef]
- Lin, T.; Liu, G.A.; Perez, E.; Rainer, R.D.; Febo, M.; Cruz-Almeida, Y.; Ebner, N.C. Systemic Inflammation Mediates Age-Related Cognitive Deficits. Front. Aging Neurosci. 2018, 10, 236. [Google Scholar] [CrossRef] [Green Version]
- Ganea, D.; Hooper, K.M.; Kong, W. The neuropeptide vasoactive intestinal peptide: Direct effects on immune cells and involvement in inflammatory and autoimmune diseases. Acta Physiol. 2015, 213, 442–452. [Google Scholar] [CrossRef] [Green Version]
- Seillet, C.; Luong, K.; Tellier, J.; Jacquelot, N.; Shen, R.D.; Hickey, P.; Wimmer, V.C.; Whitehead, L.; Rogers, K.; Smyth, G.K.; et al. The neuropeptide VIP confers anticipatory mucosal immunity by regulating ILC3 activity. Nat. Immunol. 2020, 21, 168–177. [Google Scholar] [CrossRef]
- Martínez, C.; Juarranz, Y.; Gutiérrez-Cañas, I.; Carrión, M.; Pérez-García, S.; Villanueva-Romero, R.; Castro, D.; Lamana, A.; Mellado, M.; González-Álvaro, I.; et al. A Clinical Approach for the Use of VIP Axis in Inflammatory and Autoimmune Diseases. Int. J. Mol. Sci. 2019, 21, 65. [Google Scholar] [CrossRef] [Green Version]
- Gozes, I.; Bardea, A.; Reshef, A.; Zamostiano, R.; Zhukovsky, S.; Rubinraut, S.; Fridkin, M.; Brenneman, D.E. Neuroprotective strategy for Alzheimer disease: Intranasal administration of a fatty neuropeptide. Proc. Natl. Acad. Sci. USA 1996, 93, 427–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pain, S.; Vergote, J.; Gulhan, Z.; Bodard, S.; Chalon, S.; Gaillard, A. Inflammatory process in Parkinson disease: Neuroprotection by neuropeptide Y. Fundam. Clin. Pharmacol. 2019, 33, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Szőke, É.; Bálint, M.; Hetényi, C.; Markovics, A.; Elekes, K.; Pozsgai, G.; Szűts, T.; Kéri, G.; Őrfi, L.; Sándor, Z.; et al. Small molecule somatostatin receptor subtype 4 (sst4) agonists are novel anti-inflammatory and analgesic drug candidates. Neuropharmacology 2020, 178, 108198. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liu, Y.; Liu, W.; Zhou, Y.; He, H.; Lin, S. Neuropeptide Y Is an Immunomodulatory Factor: Direct and Indirect. Front. Immunol. 2020, 11, 580378. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulos, G. Mechanisms of the systemic inflammatory response. Perfusion 1999, 14, 269–277. [Google Scholar] [CrossRef]
- Bohatschek, M.; Werner, A.; Raivich, G. Systemic LPS Injection Leads to Granulocyte Influx into Normal and Injured Brain: Effects of ICAM-1 Deficiency. Exp. Neurol. 2001, 172, 137–152. [Google Scholar] [CrossRef]
- Banks, W.A.; Niehoff, M.L.; Ponzio, N.M.; Erickson, M.A.; Zalcman, S.S. Pharmacokinetics and modeling of immune cell trafficking: Quantifying differential influences of target tissues versus lymphocytes in SJL and lipopolysaccharide-treated mice. J. Neuroinflammation 2012, 9, 714. [Google Scholar] [CrossRef] [Green Version]
- Varatharaj, A.; Galea, I. The blood-brain barrier in systemic inflammation. Brain. Behav. Immun. 2017, 60, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Sendler, M.; van den Brandt, C.; Glaubitz, J.; Wilden, A.; Golchert, J.; Weiss, F.U.; Homuth, G.; De Freitas Chama, L.L.; Mishra, N.; Mahajan, U.M.; et al. NLRP3 Inflammasome Regulates Development of Systemic Inflammatory Response and Compensatory Anti-Inflammatory Response Syndromes in Mice With Acute Pancreatitis. Gastroenterology 2020, 158, 253–269.e14. [Google Scholar] [CrossRef] [Green Version]
- Hoogland, I.C.M.; Houbolt, C.; van Westerloo, D.J.; van Gool, W.A.; van de Beek, D. Systemic inflammation and microglial activation: Systematic review of animal experiments. J. Neuroinflammation 2015, 12, 114. [Google Scholar] [CrossRef] [Green Version]
- Hamasaki, M.Y.; Barbeiro, H.V.; Barbeiro, D.F.; Cunha, D.M.G.; Koike, M.K.; Machado, M.C.C.; Pinheiro da Silva, F. Neuropeptides in the brain defense against distant organ damage. J. Neuroimmunol. 2016, 290, 33–35. [Google Scholar] [CrossRef]
- Van Epps, D.E.; Kutvirt, S.L. Modulation of human neutrophil adherence by β-endorphin and met-enkephalin. J. Neuroimmunol. 1987, 15, 219–228. [Google Scholar] [CrossRef]
- Kamphuis, S.; Eriksson, F.; Kavelaars, A.; Zijlstra, J.; van de Pol, M.; Kuis, W.; Heijnen, C.J. Role of endogenous pro-enkephalin A-derived peptides in human T cell proliferation and monocyte IL-6 production. J. Neuroimmunol. 1998, 84, 53–60. [Google Scholar] [CrossRef]
- Ferreira, R.; Santos, T.; Viegas, M.; Cortes, L.; Bernardino, L.; Vieira, O.V.; Malva, J.O. Neuropeptide Y inhibits interleukin-1β-induced phagocytosis by microglial cells. J. Neuroinflammation 2011, 8, 169. [Google Scholar] [CrossRef] [Green Version]
- Perazzo, J.; Lima, C.; Heras, M.; Bardají, E.; Lopes-Ferreira, M.; Castanho, M. Neuropeptide Kyotorphin Impacts on Lipopolysaccharide-Induced Glucocorticoid-Mediated Inflammatory Response. A Molecular Link to Nociception, Neuroprotection, and Anti-Inflammatory Action. ACS Chem. Neurosci. 2017, 8, 1663–1667. [Google Scholar] [CrossRef]
- Maldonado-Ruiz, R.; Fuentes-Mera, L.; Camacho, A. Central Modulation of Neuroinflammation by Neuropeptides and Energy-Sensing Hormones during Obesity. BioMed Res. Int. 2017, 2017, 7949582. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez-Rey, E.; Delgado, M. Vasoactive intestinal peptide inhibits cycloxygenase-2 expression in activated macrophages, microglia, and dendritic cells. Brain. Behav. Immun. 2008, 22, 35–41. [Google Scholar] [CrossRef]
- Colombo, G.; Sordi, A.; Lonati, C.; Carlin, A.; Turcatti, F.; Leonardi, P.; Gatti, S.; Catania, A. Treatment with α-melanocyte stimulating hormone preserves calcium regulatory proteins in rat heart allografts. Brain Behav. Immun. 2008, 22, 817–823. [Google Scholar] [CrossRef]
- Reinke, E.; Fabry, Z. Breaking or making immunological privilege in the central nervous system: The regulation of immunity by neuropeptides. Immunol. Lett. 2006, 104, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Duarte-Neves, J.; Pereira de Almeida, L.; Cavadas, C. Neuropeptide Y (NPY) as a therapeutic target for neurodegenerative diseases. Neurobiol. Dis. 2016, 95, 210–224. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.K.; Tuominen, L.; Tuulari, J.J.; Hirvonen, J.; Parkkola, R.; Helin, S.; Salminen, P.; Nuutila, P.; Nummenmaa, L. Obesity Is Associated with Decreased μ-Opioid But Unaltered Dopamine D2 Receptor Availability in the Brain. J. Neurosci. 2015, 35, 3959–3965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Appleyard, S.M.; Hayward, M.; Young, J.I.; Butler, A.A.; Cone, R.D.; Rubinstein, M.; Low, M.J. A Role for the Endogenous Opioid β-Endorphin in Energy Homeostasis. Endocrinology 2003, 144, 1753–1760. [Google Scholar] [CrossRef] [Green Version]
- Ramabadran, K.; Bansinath, M. Glucose homeostasis and endogenous opioid peptides. Int. J. Clin. Pharmacol. 1990, 28, 89–98. [Google Scholar]
- Mejido, D.C.P.; Peny, J.A.; Vieira, M.N.N.; Ferreira, S.T.; De Felice, F.G. Insulin and leptin as potential cognitive enhancers in metabolic disorders and Alzheimer’s disease. Neuropharmacology 2020, 171, 108115. [Google Scholar] [CrossRef]
- Butler, A.A.; Havel, P.J. Adropin and insulin resistance: Integration of endocrine, circadian, and stress signals regulating glucose metabolism. Obesity 2021, 29, 1799–1801. [Google Scholar] [CrossRef]
- Prévost, G.; Arabo, A.; Le Solliec, M.-A.; Bons, J.; Picot, M.; Maucotel, J.; Berrahmoune, H.; El Mehdi, M.; Cherifi, S.; Benani, A.; et al. Neuropeptide 26RFa (QRFP) is a key regulator of glucose homeostasis and its activity is markedly altered in obese/hyperglycemic mice. Am. J. Physiol.-Endocrinol. Metab. 2019, 317, E147–E157. [Google Scholar] [CrossRef]
- El-Mehdi, M.; Takhlidjt, S.; Khiar, F.; Prévost, G.; do Rego, J.-L.; do Rego, J.-C.; Benani, A.; Nedelec, E.; Godefroy, D.; Arabo, A.; et al. Glucose homeostasis is impaired in mice deficient in the neuropeptide 26RFa (QRFP). BMJ Open Diabetes Res. Care 2020, 8, e000942. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Chen, S.; Shen, T.; Yang, W.; Chen, Q.; Zhang, P.; You, Y.; Sun, X.; Xu, H.; Tang, Y.; et al. Adropin regulates hepatic glucose production via PP2A/AMPK pathway in insulin-resistant hepatocytes. FASEB J. 2020, 34, 10056–10072. [Google Scholar] [CrossRef]
- Banerjee, S.; Ghoshal, S.; Girardet, C.; DeMars, K.M.; Yang, C.; Niehoff, M.L.; Nguyen, A.D.; Jayanth, P.; Hoelscher, B.A.; Xu, F.; et al. Adropin correlates with aging-related neuropathology in humans and improves cognitive function in aging mice. NPJ Aging Mech. Dis. 2021, 7, 23. [Google Scholar] [CrossRef]
- Zagorácz, O.; Ollmann, T.; Péczely, L.; László, K.; Kovács, A.; Berta, B.; Kállai, V.; Kertes, E.; Lénárd, L. QRFP administration into the medial hypothalamic nuclei improves memory in rats. Brain Res. 2020, 1727, 146563. [Google Scholar] [CrossRef]
- Seminara, R.S.; Jeet, C.; Biswas, S.; Kanwal, B.; Iftikhar, W.; Sakibuzzaman, M.; Rutkofsky, I.H. The Neurocognitive Effects of Ghrelin-induced Signaling on the Hippocampus: A Promising Approach to Alzheimer’s Disease. Cureus 2018, 10, e3285. [Google Scholar] [CrossRef] [Green Version]
- Ge, X.; Yang, H.; Bednarek, M.A.; Galon-Tilleman, H.; Chen, P.; Chen, M.; Lichtman, J.S.; Wang, Y.; Dalmas, O.; Yin, Y.; et al. LEAP2 Is an Endogenous Antagonist of the Ghrelin Receptor. Cell Metab. 2018, 27, 461–469.e6. [Google Scholar] [CrossRef] [Green Version]
- Tschöp, M.; Weyer, C.; Tataranni, P.A.; Devanarayan, V.; Ravussin, E.; Heiman, M.L. Circulating Ghrelin Levels Are Decreased in Human Obesity. Diabetes 2001, 50, 707–709. [Google Scholar] [CrossRef] [Green Version]
- Makris, M.C.; Alexandrou, A.; Papatsoutsos, E.G.; Malietzis, G.; Tsilimigras, D.I.; Guerron, A.D.; Moris, D. Ghrelin and Obesity: Identifying Gaps and Dispelling Myths. A Reappraisal. Vivo Athens Greece 2017, 31, 1047–1050. [Google Scholar] [CrossRef] [Green Version]
- Mani, B.K.; Puzziferri, N.; He, Z.; Rodriguez, J.A.; Osborne-Lawrence, S.; Metzger, N.P.; Chhina, N.; Gaylinn, B.; Thorner, M.O.; Thomas, E.L.; et al. LEAP2 changes with body mass and food intake in humans and mice. J. Clin. Investig. 2019, 129, 3909–3923. [Google Scholar] [CrossRef]
- Belsham, D.D.; Dalvi, P.S. Insulin signalling in hypothalamic neurones. J. Neuroendocrinol. 2021, 33, e12919. [Google Scholar] [CrossRef]
- Brent, A.E.; Rajan, A. Insulin and Leptin/Upd2 Exert Opposing Influences on Synapse Number in Fat-Sensing Neurons. Cell Metab. 2020, 32, 786–800.e7. [Google Scholar] [CrossRef]
- Manuel, I.; Lombardero, L.; Llorente-Ovejero, A.; Rodríguez-Puertas, R. Neuropeptides and neurolipids. In Genetics, Neurology, Behavior, and Diet in Dementia; Elsevier: Amsterdam, The Netherlands, 2020; pp. 423–439. ISBN 978-0-12-815868-5. [Google Scholar]
- Elvander, E.; Ögren, S.O. Medial septal galanin and acetylcholine: Influence on hippocampal acetylcholine and spatial learning. Neuropeptides 2005, 39, 245–248. [Google Scholar] [CrossRef]
- Dutar, P.; Vaillend, C.; Viollet, C.; Billard, J.-M.; Potier, B.; Carlo, A.-S.; Ungerer, A.; Epelbaum, J. Spatial learning and synaptic hippocampal plasticity in type 2 somatostatin receptor knock-out mice. Neuroscience 2002, 112, 455–466. [Google Scholar] [CrossRef]
- Hupalo, S.; Bryce, C.A.; Bangasser, D.A.; Berridge, C.W.; Valentino, R.J.; Floresco, S.B. Corticotropin-Releasing Factor (CRF) circuit modulation of cognition and motivation. Neurosci. Biobehav. Rev. 2019, 103, 50–59. [Google Scholar] [CrossRef]
- Whissell, P.D.; Bang, J.Y.; Khan, I.; Xie, Y.-F.; Parfitt, G.M.; Grenon, M.; Plummer, N.W.; Jensen, P.; Bonin, R.P.; Kim, J.C. Selective Activation of Cholecystokinin-Expressing GABA (CCK-GABA) Neurons Enhances Memory and Cognition. Eneuro 2019, 6, ENEURO.0360-18.2019. [Google Scholar] [CrossRef]
- Selkoe, D.J.; Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 2016, 8, 595–608. [Google Scholar] [CrossRef]
- Ishii, M.; Hiller, A.J.; Pham, L.; McGuire, M.J.; Iadecola, C.; Wang, G. Amyloid-Beta Modulates Low-Threshold Activated Voltage-Gated L-Type Calcium Channels of Arcuate Neuropeptide Y Neurons Leading to Calcium Dysregulation and Hypothalamic Dysfunction. J. Neurosci. 2019, 39, 8816–8825. [Google Scholar] [CrossRef]
- Li, C.; Wu, X.; Liu, S.; Zhao, Y.; Zhu, J.; Liu, K. Roles of Neuropeptide Y in Neurodegenerative and Neuroimmune Diseases. Front. Neurosci. 2019, 13, 869. [Google Scholar] [CrossRef]
- Chen, X.-Y.; Du, Y.-F.; Chen, L. Neuropeptides Exert Neuroprotective Effects in Alzheimer’s Disease. Front. Mol. Neurosci. 2019, 11, 493. [Google Scholar] [CrossRef] [Green Version]
- Pain, S.; Brot, S.; Gaillard, A. Neuroprotective effects of neuropeptide Y against neurodegenerative disease. Curr. Neuropharmacol. 2021, 19. [Google Scholar] [CrossRef]
- Clark, C.M.; Clark, R.M.; Hoyle, J.A.; Dickson, T.C. Pathogenic or protective? Neuropeptide Y in amyotrophic lateral sclerosis. J. Neurochem. 2021, 156, 273–289. [Google Scholar] [CrossRef]
- Patil, M.J.; Henry, M.A.; Akopian, A.N. Prolactin receptor in regulation of neuronal excitability and channels. Channels 2014, 8, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, H.D.; Yu, B.P.; Hoang, N.H.M.; Jo, W.H.; Young Chung, H.; Kim, M.-S. Prolactin and its altered action in Alzheimer’s disease and Parkinson’s disease. Neuroendocrinology 2021. [Google Scholar] [CrossRef] [PubMed]
- Al-Ahmad, A.J.; Pervaiz, I.; Karamyan, V.T. Neurolysin substrates bradykinin, neurotensin and substance P enhance brain microvascular permeability in a human in vitro model. J. Neuroendocrinol. 2021, 33, e12931. [Google Scholar] [CrossRef] [PubMed]
- Nozohouri, S.; Jayaraman, S.; Vaidya, B.; Karamyan, V.; Abbruscato, T. Anti-edematous effect of neurolysin in ischemic stroke. FASEB J. 2019, 33, lb82. [Google Scholar] [CrossRef]
- Kim, N.S.; Oh, S.-M.; Ko, M.M.; Cha, M.H.; Kang, B.K.; Bang, O.S. Association of the C-399T promoter polymorphism of neuropeptide Y with susceptibility to ischemic stroke. Clin. Biochem. 2009, 42, 1699–1704. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-T.; Yu, N.-N.; Gao, S.-S.; Song, J.-H.; Ma, T.; Wang, N.-D.; Tang, Y.-C.; Zhang, N.; Tan, L. Neuropeptide Y polymorphisms and ischemic stroke in Chinese population. Clin. Chim. Acta 2010, 411, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.S.; Ko, M.M.; Cha, M.H.; Oh, S.-M.; Bang, O.S. Age and sex dependent genetic effects of neuropeptide Y promoter polymorphism on susceptibility to ischemic stroke in Koreans. Clin. Chim. Acta 2010, 411, 1243–1247. [Google Scholar] [CrossRef]
- Sun, W.; Zhang, Z.; Feng, X.; Sui, X.; Miao, Y. Serum Neuropeptide Y: A Potential Prognostic Marker of Intracerebral Hemorrhage. Dis. Markers 2021, 2021, 7957013. [Google Scholar] [CrossRef]
- Wang, N.; Wang, D.; Zhou, H.; Xu, C.; Hu, X.; Qian, Z.; Xu, X. Serum Neuropeptide Y Level is Associated with Post-Ischemic Stroke Epilepsy. J. Stroke Cerebrovasc. Dis. 2021, 30, 105475. [Google Scholar] [CrossRef]
- Werner, F.-M.; Covenas, R. Classical Neurotransmitters and Neuropeptides Involved in Generalized Epilepsy: A Focus on Antiepileptic Drugs. Curr. Med. Chem. 2011, 18, 4933–4948. [Google Scholar] [CrossRef]
- Govardhanagiri, S.; Bethi, S.; Nagaraju, G.P. Chapter 8—Small Molecules and Pancreatic Cancer Trials and Troubles. In Breaking Tolerance to Pancreatic Cancer Unresponsiveness to Chemotherapy; Nagaraju, G.P., Ed.; Cancer Sensitizing Agents for Chemotherapy; Academic Press: Cambridge, MA, USA, 2019; Volume 5, pp. 117–131. [Google Scholar]
- Casey, D.A.; Antimisiaris, D.; O’Brien, J. Drugs for Alzheimer’s Disease: Are They Effective? Pharm. Ther. 2010, 35, 208–211. [Google Scholar]
- Roy, S.; Kundu, T.K. II—Methods: Tools for studying genomes and gene functions in intact cells. In Chemical Biology of the Genome; Roy, S., Kundu, T.K., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 27–73. ISBN 978-0-12-817644-3. [Google Scholar]
- Kim, D.-K.; Cho, E.B.; Moon, M.J.; Park, S.; Hwang, J.-I.; Do Rego, J.-L.; Vaudry, H.; Seong, J.Y. Molecular Coevolution of Neuropeptides Gonadotropin-Releasing Hormone and Kisspeptin with their Cognate G Protein-Coupled Receptors. Front. Neurosci. 2012, 6, 3. [Google Scholar] [CrossRef] [Green Version]
- Sahu, B.S.; Rodriguez, P.; Nguyen, M.E.; Han, R.; Cero, C.; Razzoli, M.; Piaggi, P.; Laskowski, L.J.; Pavlicev, M.; Muglia, L.; et al. Peptide/Receptor Co-evolution Explains the Lipolytic Function of the Neuropeptide TLQP-21. Cell Rep. 2019, 28, 2567–2580.e6. [Google Scholar] [CrossRef] [Green Version]
- Salio, C.; Averill, S.; Priestley, J.V.; Merighi, A. Costorage of BDNF and neuropeptides within individual dense-core vesicles in central and peripheral neurons. Dev. Neurobiol. 2007, 67, 326–338. [Google Scholar] [CrossRef]
- Bhat, U.S.; Shahi, N.; Surendran, S.; Babu, K. Neuropeptides and Behaviors: How Small Peptides Regulate Nervous System Function and Behavioral Outputs. Front. Mol. Neurosci. 2021, 14, 306. [Google Scholar] [CrossRef]
- Cabri, W.; Cantelmi, P.; Corbisiero, D.; Fantoni, T.; Ferrazzano, L.; Martelli, G.; Mattellone, A.; Tolomelli, A. Therapeutic Peptides Targeting PPI in Clinical Development: Overview, Mechanism of Action and Perspectives. Front. Mol. Biosci. 2021, 8, 697586. [Google Scholar] [CrossRef]
- Coin, I.; Beyermann, M.; Bienert, M. Solid-phase peptide synthesis: From standard procedures to the synthesis of difficult sequences. Nat. Protoc. 2007, 2, 3247–3256. [Google Scholar] [CrossRef]
- Gaglione, R.; Pane, K.; Dell’Olmo, E.; Cafaro, V.; Pizzo, E.; Olivieri, G.; Notomista, E.; Arciello, A. Cost-effective production of recombinant peptides in Escherichia coli. New Biotechnol. 2019, 51, 39–48. [Google Scholar] [CrossRef]
- Chen, R. Bacterial expression systems for recombinant protein production: E. coli and beyond. Biotechnol. Adv. 2012, 30, 1102–1107. [Google Scholar] [CrossRef]
- Vecchio, I.; Tornali, C.; Bragazzi, N.L.; Martini, M. The Discovery of Insulin: An Important Milestone in the History of Medicine. Front. Endocrinol. 2018, 9, 613. [Google Scholar] [CrossRef] [Green Version]
- Noren, C.J.; Anthony-Cahill, S.J.; Griffith, M.C.; Schultz, P.G. A General Method for Site-specific Incorporation of Unnatural Amino Acids into Proteins. Science 1989, 244, 182–188. [Google Scholar] [CrossRef] [Green Version]
- Van Vught, R.; Pieters, R.J.; Breukink, E. Site-specific functionalization of proteins and their applications to therapeutic antibodies. Comput. Struct. Biotechnol. J. 2014, 9, e201402001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pawlas, J.; Nuijens, T.; Persson, J.; Svensson, T.; Schmidt, M.; Toplak, A.; Nilsson, M.; Rasmussen, J.H. Sustainable, cost-efficient manufacturing of therapeutic peptides using chemo-enzymatic peptide synthesis (CEPS). Green Chem. 2019, 21, 6451–6467. [Google Scholar] [CrossRef]
- Hamman, J.H.; Enslin, G.M.; Kotz??, A.F. Oral Delivery of Peptide Drugs: Barriers and Developments. BioDrugs 2005, 19, 165–177. [Google Scholar] [CrossRef] [PubMed]
- John, H.; Maronde, E.; Forssmann, W.-G.; Meyer, M.; Adermann, K. N-terminal acetylation protects glucagon-like peptide GLP-1-(7-34)-amide from DPP-IV-mediated degradation retaining cAMP- and insulin-releasing capacity. Eur. J. Med. Res. 2008, 13, 73–78. [Google Scholar]
- Räder, A.F.B.; Reichart, F.; Weinmüller, M.; Kessler, H. Improving oral bioavailability of cyclic peptides by N-methylation. Bioorg. Med. Chem. 2018, 26, 2766–2773. [Google Scholar] [CrossRef]
- Sharma, A.; Kumar, A.; Abdel Monaim, S.A.H.; Jad, Y.E.; El-Faham, A.; de la Torre, B.G.; Albericio, F. N -methylation in amino acids and peptides: Scope and limitations. Biopolymers 2018, 109, e23110. [Google Scholar] [CrossRef]
- Partridge, A.W.; Kaan, H.Y.K.; Juang, Y.-C.; Sadruddin, A.; Lim, S.; Brown, C.J.; Ng, S.; Thean, D.; Ferrer, F.; Johannes, C.; et al. Incorporation of Putative Helix-Breaking Amino Acids in the Design of Novel Stapled Peptides: Exploring Biophysical and Cellular Permeability Properties. Molecules 2019, 24, 2292. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Mietlicki-Baase, E.G.; Barrett, T.M.; McGrath, L.E.; Koch-Laskowski, K.; Ferrie, J.J.; Hayes, M.R.; Petersson, E.J. Thioamide Substitution Selectively Modulates Proteolysis and Receptor Activity of Therapeutic Peptide Hormones. J. Am. Chem. Soc. 2017, 139, 16688–16695. [Google Scholar] [CrossRef]
- Dohm, M.T.; Kapoor, R.; Barron, A.E. Peptoids: Bio-inspired polymers as potential pharmac Peptoids: Bio-Inspired Polymers as Potential Pharmaceuticals. Curr. Pharm. Des. 2011, 17, 2732–2747. [Google Scholar] [CrossRef] [Green Version]
- Hayes, H.C.; Luk, L.Y.P.; Tsai, Y.-H. Approaches for peptide and protein cyclisation. Org. Biomol. Chem. 2021, 19, 3983–4001. [Google Scholar] [CrossRef]
- Gori, A.; Gagni, P.; Rinaldi, S. Disulfide Bond Mimetics: Strategies and Challenges. Chem. Eur. J. 2017, 23, 14987–14995. [Google Scholar] [CrossRef]
- Wu, H.; Huang, J. Optimization of Protein and Peptide Drugs Based on the Mechanisms of Kidney Clearance. Protein Pept. Lett. 2018, 25, 514–521. [Google Scholar] [CrossRef]
- Bak, M.; Park, J.; Min, K.; Cho, J.; Seong, J.; Hahn, Y.S.; Tae, G.; Kwon, I. Recombinant Peptide Production Platform Coupled with Site-Specific Albumin Conjugation Enables a Convenient Production of Long-Acting Therapeutic Peptide. Pharmaceutics 2020, 12, 364. [Google Scholar] [CrossRef]
- Zorzi, A.; Middendorp, S.J.; Wilbs, J.; Deyle, K.; Heinis, C. Acylated heptapeptide binds albumin with high affinity and application as tag furnishes long-acting peptides. Nat. Commun. 2017, 8, 16092. [Google Scholar] [CrossRef]
- Kugler, A.; Thiman, M. Efficacy and safety profile of once-weekly dulaglutide in type 2 diabetes: A report on the emerging new data. Diabetes Metab. Syndr. Obes. Targets Ther. 2018, 11, 187–197. [Google Scholar] [CrossRef] [Green Version]
- Asfour, M.H. Advanced trends in protein and peptide drug delivery: A special emphasis on aquasomes and microneedles techniques. Drug Deliv. Transl. Res. 2021, 11, 1–23. [Google Scholar] [CrossRef]
- Han, Y.; Gao, Z.; Chen, L.; Kang, L.; Huang, W.; Jin, M.; Wang, Q.; Bae, Y.H. Multifunctional oral delivery systems for enhanced bioavailability of therapeutic peptides/proteins. Acta Pharm. Sin. B 2019, 9, 902–922. [Google Scholar] [CrossRef]
- Eldor, R.; Neutel, J.; Homer, K.; Kidron, M. Efficacy and safety of 28-day treatment with oral insulin (ORMD-0801) in patients with type 2 diabetes: A randomized, placebo-controlled trial. Diabetes Obes. Metab. 2021, 23, 2529–2538. [Google Scholar] [CrossRef]
- Pandit, R.; Chen, L.; Götz, J. The blood-brain barrier: Physiology and strategies for drug delivery. Adv. Drug Deliv. Rev. 2020, 165–166, 1–14. [Google Scholar] [CrossRef]
- Sweeney, M.D.; Zhao, Z.; Montagne, A.; Nelson, A.R.; Zlokovic, B.V. Blood-Brain Barrier: From Physiology to Disease and Back. Physiol. Rev. 2019, 99, 21–78. [Google Scholar] [CrossRef]
- Sanchez-Covarrubias, L.; Slosky, L.; Thompson, B.; Davis, T.; Ronaldson, P. Transporters at CNS Barrier Sites: Obstacles or Opportunities for Drug Delivery? Curr. Pharm. Des. 2014, 20, 1422–1449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.-P.; Ahmadvand, D.; Su, J.; Hall, A.; Tan, X.; Farhangrazi, Z.S.; Moghimi, S.M. Crossing the blood-brain-barrier with nanoligand drug carriers self-assembled from a phage display peptide. Nat. Commun. 2019, 10, 4635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arranz-Gibert, P.; Prades, R.; Guixer, B.; Guerrero, S.; Araya, E.; Ciudad, S.; Kogan, M.J.; Giralt, E.; Teixidó, M. HAI Peptide and Backbone Analogs—Validation and Enhancement of Biostability and Bioactivity of BBB Shuttles. Sci. Rep. 2018, 8, 17932. [Google Scholar] [CrossRef] [PubMed]
- Vinzant, N.; Scholl, J.L.; Wu, C.-M.; Kindle, T.; Koodali, R.; Forster, G.L. Iron Oxide Nanoparticle Delivery of Peptides to the Brain: Reversal of Anxiety during Drug Withdrawal. Front. Neurosci. 2017, 11, 608. [Google Scholar] [CrossRef] [Green Version]
- Spencer, B.; Potkar, R.; Metcalf, J.; Thrin, I.; Adame, A.; Rockenstein, E.; Masliah, E. Systemic Central Nervous System (CNS)-targeted Delivery of Neuropeptide Y (NPY) Reduces Neurodegeneration and Increases Neural Precursor Cell Proliferation in a Mouse Model of Alzheimer Disease. J. Biol. Chem. 2016, 291, 1905–1920. [Google Scholar] [CrossRef] [Green Version]
- Zaman, R.U.; Mulla, N.S.; Braz Gomes, K.; D’Souza, C.; Murnane, K.S.; D’Souza, M.J. Nanoparticle formulations that allow for sustained delivery and brain targeting of the neuropeptide oxytocin. Int. J. Pharm. 2018, 548, 698–706. [Google Scholar] [CrossRef]
- Erdő, F.; Bors, L.A.; Farkas, D.; Bajza, Á.; Gizurarson, S. Evaluation of intranasal delivery route of drug administration for brain targeting. Brain Res. Bull. 2018, 143, 155–170. [Google Scholar] [CrossRef]
- Benedict, C.; Hallschmid, M.; Schmitz, K.; Schultes, B.; Ratter, F.; Fehm, H.L.; Born, J.; Kern, W. Intranasal Insulin Improves Memory in Humans: Superiority of Insulin Aspart. Neuropsychopharmacology 2007, 32, 239–243. [Google Scholar] [CrossRef] [Green Version]
- Craft, S.; Claxton, A.; Baker, L.D.; Hanson, A.J.; Cholerton, B.; Trittschuh, E.H.; Dahl, D.; Caulder, E.; Neth, B.; Montine, T.J.; et al. Effects of Regular and Long-Acting Insulin on Cognition and Alzheimer’s Disease Biomarkers: A Pilot Clinical Trial. J. Alzheimers Dis. JAD 2017, 57, 1325–1334. [Google Scholar] [CrossRef] [Green Version]
- Shin, N.Y.; Park, H.Y.; Jung, W.H.; Park, J.W.; Yun, J.-Y.; Jang, J.H.; Kim, S.N.; Han, H.J.; Kim, S.-Y.; Kang, D.-H.; et al. Effects of Oxytocin on Neural Response to Facial Expressions in Patients with Schizophrenia. Neuropsychopharmacology 2015, 40, 1919–1927. [Google Scholar] [CrossRef]
- Van Zuiden, M.; Frijling, J.L.; Nawijn, L.; Koch, S.B.J.; Goslings, J.C.; Luitse, J.S.; Biesheuvel, T.H.; Honig, A.; Veltman, D.J.; Olff, M. Intranasal Oxytocin to Prevent Posttraumatic Stress Disorder Symptoms: A Randomized Controlled Trial in Emergency Department Patients. Biol. Psychiatry 2017, 81, 1030–1040. [Google Scholar] [CrossRef] [Green Version]
- Parker, K.J.; Oztan, O.; Libove, R.A.; Sumiyoshi, R.D.; Jackson, L.P.; Karhson, D.S.; Summers, J.E.; Hinman, K.E.; Motonaga, K.S.; Phillips, J.M.; et al. Intranasal oxytocin treatment for social deficits and biomarkers of response in children with autism. Proc. Natl. Acad. Sci. USA 2017, 114, 8119–8124. [Google Scholar] [CrossRef] [Green Version]
- Lovejoy, D.A.; Hogg, D.W.; Dodsworth, T.L.; Jurado, F.R.; Read, C.C.; D’Aquila, A.L.; Barsyte-Lovejoy, D. Synthetic Peptides as Therapeutic Agents: Lessons Learned From Evolutionary Ancient Peptides and Their Transit Across Blood-Brain Barriers. Front. Endocrinol. 2019, 10, 730. [Google Scholar] [CrossRef]
- Basith, S.; Manavalan, B.; Hwan Shin, T.; Lee, G. Machine intelligence in peptide therapeutics: A next-generation tool for rapid disease screening. Med. Res. Rev. 2020, 40, 1276–1314. [Google Scholar] [CrossRef]
- Mulligan, V.K. The emerging role of computational design in peptide macrocycle drug discovery. Expert Opin. Drug Discov. 2020, 15, 833–852. [Google Scholar] [CrossRef]
- Sindhikara, D.; Wagner, M.; Gkeka, P.; Güssregen, S.; Tiwari, G.; Hessler, G.; Yapici, E.; Li, Z.; Evers, A. Automated Design of Macrocycles for Therapeutic Applications: From Small Molecules to Peptides and Proteins. J. Med. Chem. 2020, 63, 12100–12115. [Google Scholar] [CrossRef]
- Tanday, N.; Flatt, P.R.; Irwin, N. Amplifying the antidiabetic actions of glucagon-like peptide-1: Potential benefits of new adjunct therapies. Diabet. Med. 2021, 38, e14699. [Google Scholar] [CrossRef]
- Baggio, L.L.; Drucker, D.J. Glucagon-like peptide-1 receptor co-agonists for treating metabolic disease. Mol. Metab. 2021, 46, 101090. [Google Scholar] [CrossRef]
- Merkel, R.; Moreno, A.; Zhang, Y.; Herman, R.; Ben Nathan, J.; Zeb, S.; Rahematpura, S.; Stecyk, K.; Milliken, B.T.; Hayes, M.R.; et al. A novel approach to treating opioid use disorders: Dual agonists of glucagon-like peptide-1 receptors and neuropeptide Y2 receptors. Neurosci. Biobehav. Rev. 2021, 131, 1169–1179. [Google Scholar] [CrossRef]
- Muchowska, A.; Redkiewicz, P.; Różycki, K.; Matalińska, J.; Lipiński, P.F.J.; Czuwara, J.; Kosson, P. The analgesic hybrid of dermorphin/substance P and analog of enkephalin improve wound healing in streptozotocin-induced diabetic rats. Wound Repair Regen. 2020, 28, 177–184. [Google Scholar] [CrossRef]
- Bądzyńska, B.; Lipkowski, A.W.; Sadowski, J. An antihypertensive opioid: Biphalin, a synthetic non-addictive enkephalin analog decreases blood pressure in spontaneously hypertensive rats. Pharmacol. Rep. 2016, 68, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Sotgiu, M.L.; Valente, M.; Storchi, R.; Caramenti, G.; Biella, G.E.M. Cooperative N-methyl-d-aspartate (NMDA) receptor antagonism and μ-opioid receptor agonism mediate the methadone inhibition of the spinal neuron pain-related hyperactivity in a rat model of neuropathic pain. Pharmacol. Res. 2009, 60, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Palmer, C.B.; Meyrath, M.; Canals, M.; Kostenis, E.; Chevigné, A.; Szpakowska, M. Atypical opioid receptors: Unconventional biology and therapeutic opportunities. Pharmacol. Ther. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, S.; Verlengia, G.; Marino, P.; Simonato, M.; Bettegazzi, B. NPY and Gene Therapy for Epilepsy: How, When,…and Y. Front. Mol. Neurosci. 2021, 13, 608001. [Google Scholar] [CrossRef]
- Marlin, B.J.; Froemke, R.C. Oxytocin modulation of neural circuits for social behavior: Oxytocin Modulation of Neural Circuits for Social Behavior. Dev. Neurobiol. 2017, 77, 169–189. [Google Scholar] [CrossRef]
- Ciranna, L.; Costa, L. Pituitary Adenylate Cyclase-Activating Polypeptide Modulates Hippocampal Synaptic Transmission and Plasticity: New Therapeutic Suggestions for Fragile X Syndrome. Front. Cell. Neurosci. 2019, 13, 524. [Google Scholar] [CrossRef]
- Yang, R.; Jiang, X.; Ji, R.; Meng, L.; Liu, F.; Chen, X.; Xin, Y. Therapeutic potential of PACAP for neurodegenerative diseases. Cell. Mol. Biol. Lett. 2015, 20, 265–278. [Google Scholar] [CrossRef]
- Cabezas-Llobet, N.; Vidal-Sancho, L.; Masana, M.; Fournier, A.; Alberch, J.; Vaudry, D.; Xifró, X. Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) Enhances Hippocampal Synaptic Plasticity and Improves Memory Performance in Huntington’s Disease. Mol. Neurobiol. 2018, 55, 8263–8277. [Google Scholar] [CrossRef]
- Li, C.; Li, M.; Yu, H.; Shen, X.; Wang, J.; Sun, X.; Wang, Q.; Wang, C. Neuropeptide VGF C-Terminal Peptide TLQP-62 Alleviates Lipopolysaccharide-Induced Memory Deficits and Anxiety-like and Depression-like Behaviors in Mice: The Role of BDNF/TrkB Signaling. ACS Chem. Neurosci. 2017, 8, 2005–2018. [Google Scholar] [CrossRef]
- Johnson, M.B.; Young, A.D.; Marriott, I. The Therapeutic Potential of Targeting Substance P/NK-1R Interactions in Inflammatory CNS Disorders. Front. Cell. Neurosci. 2017, 10, 296. [Google Scholar] [CrossRef] [Green Version]
- Krishnaswamy, R.; Malik, B.H.; Khan, S.; Gupta, D.; Islam, M.; Mandal, S.K.; Rutkofsky, I.H. Anti-CGRP monoclonal antibodies: Breakthrough in migraine therapeutics. Prog. Neurol. Psychiatry 2019, 23, 26–33. [Google Scholar] [CrossRef] [Green Version]
- Lerner, J.T.; Sankar, R.; Mazarati, A.M. Galanin—25 years with a multitalented neuropeptide. Cell. Mol. Life Sci. 2008, 65, 1864–1871. [Google Scholar] [CrossRef]
- Henry, M.S.; Gendron, L.; Tremblay, M.-E.; Drolet, G. Enkephalins: Endogenous Analgesics with an Emerging Role in Stress Resilience. Neural Plast. 2017, 2017, 1546125. [Google Scholar] [CrossRef]
- Boniakowski, A.E.; Kimball, A.S.; Jacobs, B.N.; Kunkel, S.L.; Gallagher, K.A. Macrophage-Mediated Inflammation in Normal and Diabetic Wound Healing. J. Immunol. 2017, 199, 17–24. [Google Scholar] [CrossRef] [Green Version]

| Peptide | Trade Name | Target Condition | Modifications to the Original Structure | Year of Approval | Remarks |
|---|---|---|---|---|---|
| Vasopressin | Desmopressin | Diabetes insipidus [13], nocturia [14] | No modifications | Diabetes insipidus in 1978, nocturia in 2017. | Purified posterior pituitary extract was used before the synthetic production of vasopressin [15]. |
| Oxytocin | Pitocin | Obstetrics, to induce labor and prevent postpartum bleeding [16] | No modifications | 1980 | First peptide hormone synthesized in the lab [17]. |
| Insulin [10] | Insulin lispro, insulin aspart, insulin glulisine (rapid-acting), insulin glargine, insulin detemir, insulin degludec (long-acting) | Type I and II diabetes mellitus | Single amino acid modifications increase speed of release [18]. Insulin determir and degludec conjugated with fatty acids to enhance albumin binding, improving pharmacokinetics [19,20]. | First analogue (insulin lispro) approved in 1982. | Alternative delivery methods attempted: implantable device, needle-free jet injection, transdermal delivery, microneedle-based delivery, pulmonary delivery [21,22,23,24,25]. There has been a shift in reliance, from synthetic human insulin towards insulin analogues. In 2010, 91.5% and 14.8% of type II diabetes patients use analogues and human synthetic peptides respectively, compared to 18.6% and 96.4% in 2000 [26]. |
| Parathyroid hormone [27,28] | Teriparatide | Osteoporosis | 34 amino acids in the N terminus is used. | 2002 | Designed using recombinant technology. |
| Calcitonin [29] | Fortical (recombinant salmon calcitonin) | Hypercalcaemia, postmenopausal osteoporosis | No modifications | 2005 | Designed using recombinant technology. Restricted use due to increased cancer risk [30]. |
| Exendin-4 [31] | Exenatide | Type II diabetes mellitus | Synthetic version of exendin-4. | 2005 | Glucagon-like peptide 1 (GLP-1) receptor agonist. Synthesized on gram scale via enzymatic ligation of synthetic peptide fragments. |
| Adrenocortico-tropic hormone (ACTH) | Achtar gel | Infantile spasms [32] | No modifications | 2010 | ACTH initially isolated and introduced to treat endocrine disorders in the 1950s [10]. |
| GLP-1 | Liraglutide, semaglutide (Rybelsus) | Type II diabetes mellitus, obesity | Conjugated with fatty acids in order to enhance binding to albumin, improving pharmacokinetics [33,34]. | Liraglutide in 2014, semaglutide in 2019. | Glucagon-like peptide-1 analogue [34]. |
| Somatostatin [35] | Octreotide | Acromegaly, diarrhea associated with metastatic carcinoid tumors and VIP-secreting tumors | Encapsulated with proprietary excipients (transient permeability enhancer). | 2020 | - |
| Difelikefalin [36] | Korsuva | Moderate to severe itching associated with chronic kidney disease | No modifications | 2021 | Κ-opioid receptor agonist. |
| Peptide/s | Possible Targets and Mechanisms of Action | Systems Affected | Potential Application |
|---|---|---|---|
| NPY | Neuroprotective [211], regulation of immune cell function [145]. | Widely expressed in the peripheral system and the central nervous system, including the hippocampus, hypothalamus, amygdala, striatum. | Wide range of neurological disorders, including AD, PD, HD, Machado-Joseph disease [186,211,212], ALS [214], as well as associated inflammatory processes [168,169]. Potential anticonvulsant effects against seizures [281]. |
| Oxytocin | Modulation of LTP and LTD of synapses during early development [66,105,282]. Suggested role in early stages of the systemic inflammatory response [177]. | Predominant synthesis and expression in the hypothalamus. Expressed in lower densities brain wide. | Schizophrenia [267], post-traumatic stress disorder [268], and ASD [61,65,269]. |
| Adropin and neuropeptide 26RFa | Regulate insulin and glucose homeostasis and cardiovascular function in the periphery [133,191,192,193,194]. Reported in the maintenance of neuronal homeostasis during aging [195,196]. | Peripheral tissues associated with metabolic control and energy homeostasis. Hypothalamus, possibly hippocampus. | Metabolic dysfunction associated with obesity. Cognitive dysfunction as a result of aging. |
| LEAP-2 and ghrelin | Balance of ghrelin antagonism by LEAP-2 not only controls food intake [198,199,200], but has also been suggested to regulate spatial learning and memory [195,196]. | Peripheral tissues associated with metabolic control. Hippocampus. | Obesity, learning, and memory problems associated with AD. |
| Prolactin | Reported roles in neurogenesis and neuronal stem cell proliferation. Expressed on microglia and astrocytes with suggestive roles in inflammatory response [215]. | Hypothalamus, hippocampus, cortex. | AD, PD [216]. |
| PACAP | Regulates synaptic plasticity via the modulation of glutamatergic transmission during development [68] and adulthood [283]. Reported role in immune response receptors expressed on microglia [144]. | Widely expressed in the brain, including in the hippocampus and hypothalamus. | AD and PD [284], HD [285], Fragile X syndrome [283]. |
| TLQP-62 | Regulates developmental synaptic plasticity [69], neuroinflammatory and oxidative responses [286]. | Hippocampus. | Neuropsychiatric disorders. |
| Neurolysin | Regulates activity of other neuropeptides, control of inflammation and excitotoxicity during ischaemic stroke [218]. | Brain-wide effects. | Ischaemic stroke. |
| Nocistatin, big dynorphin and RFamide | Activate acid-sensing ion channels during stroke, facilitating acidosis and exacerbating neuronal death [86,87,88,89]. | Brain-wide effects. | Inhibiting actions of these neuropeptides has the potential to reduce ASIC activation during ischaemic stroke, which may reduce subsequent pathological and inflammatory effects [90]. |
| CGRP and substance P | Regulate inflammatory processes [144,145,146,147]. Enhance neuronal excitability underlying the response to pain [118,119,131,134]. | Brain-wide effects. Actions in the nucleus parabrachialis, BLA, and CeA are particularly important in regulation of the pain response. | Inhibiting actions of CGRP/SP may reduce pathological effects of inflammatory disorders [287] and the pain response, e.g., as a result of migraines [288]. |
| VIP and somatostatin [166,167,169] | Anti-inflammatory mediators of immune cell function. | Many peripheral locations, including GI tract, heart, kidneys, thyroid gland. Brain areas include hypothalamus, pituitary gland. | Neurological and peripheral disorders with associated inflammatory processes or autoimmunity. |
| Galanin | A primarily inhibitory role, possibly via the activation of serotonergic pathways [289]. | Widely expressed in the peripheral and central nervous system, including in the medial temporal lobe. | Epileptic seizures [224]. |
| Biphalin | An enkephalin analogue. Enkephalins have been implicated in pain responses [127,128], stress [290], and the inflammatory response [178,179]. | Enkephalins are highly expressed in the limbic system of the CNS, peripheral organs such as the skin, liver, and lungs, and the adrenal medulla [290]. | May accelerate immune system activation, a reduction of which has been associated with diabetes [277,291]. Hypertension [278]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeo, X.Y.; Cunliffe, G.; Ho, R.C.; Lee, S.S.; Jung, S. Potentials of Neuropeptides as Therapeutic Agents for Neurological Diseases. Biomedicines 2022, 10, 343. https://doi.org/10.3390/biomedicines10020343
Yeo XY, Cunliffe G, Ho RC, Lee SS, Jung S. Potentials of Neuropeptides as Therapeutic Agents for Neurological Diseases. Biomedicines. 2022; 10(2):343. https://doi.org/10.3390/biomedicines10020343
Chicago/Turabian StyleYeo, Xin Yi, Grace Cunliffe, Roger C. Ho, Su Seong Lee, and Sangyong Jung. 2022. "Potentials of Neuropeptides as Therapeutic Agents for Neurological Diseases" Biomedicines 10, no. 2: 343. https://doi.org/10.3390/biomedicines10020343
APA StyleYeo, X. Y., Cunliffe, G., Ho, R. C., Lee, S. S., & Jung, S. (2022). Potentials of Neuropeptides as Therapeutic Agents for Neurological Diseases. Biomedicines, 10(2), 343. https://doi.org/10.3390/biomedicines10020343