Nutritional Profiles, Phytochemical Analysis, Antioxidant Activity and DNA Damage Protection of Makapuno Derived from Thai Aromatic Coconut
Abstract
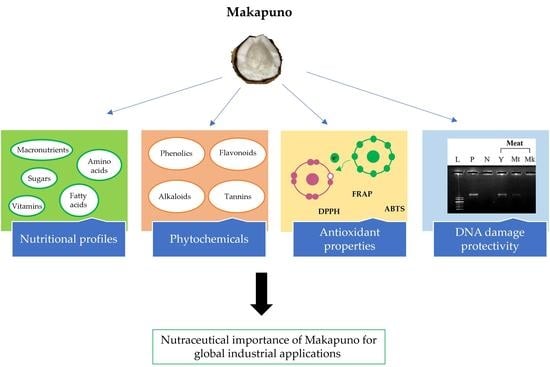
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Nutritional Value Analysis
2.3. Sample Preparations
2.3.1. Preparation of Coconut Meat and Methanolic Extracts
2.3.2. Preparation of Coconut Water
2.4. Determination of Phytochemical Constituents
2.4.1. Total Phenolic Content
2.4.2. Flavonoid Content
2.4.3. Tannin Content
2.4.4. Alkaloids
2.5. Evaluation of Antioxidant Activity
2.5.1. Determination of Antioxidant Activity Using the 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Method
2.5.2. Determination of Antioxidant Activity Using the Ferric Reducing/Antioxidant Power (FRAP) Method
2.5.3. Determination of Antioxidant Activity Using the ABTS Free Radical Scavenging Method
2.5.4. Analysis of DNA Damage Protection
2.6. Statistical Analysis
3. Results and Discussion
3.1. Nutritional Composition
3.2. Phytochemical Constituents
3.3. Evaluation of Antioxidant Activities
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Moumita, C.; Adinpunya, M. The antioxidant and antimicrobial properties of the methanolic extract from Cocos nucifera mesocarp. Food Chem. 2008, 107, 994–999. [Google Scholar] [CrossRef]
- Reddy, P.E.; Lakshmi, M.T. Coconut water- properties, uses, nutritional benefits in health and wealth and in health and disease. J. Curr. Trends Clin. Med. Lab. Biochem. 2014, 2, 618. [Google Scholar]
- Tuyekar, S.T.; Tawade, B.S.; Singh, K.S.; Wagh, V.S.; Vidhate, P.K.; Yevale, R.P.; Gaikwad, S.; Kale, M. An Overview on Coconut Water: As A Multipurpose Nutrition. Int. J. Pharm. Sci. Rev. Res. 2021, 68, 63–70. [Google Scholar] [CrossRef]
- Bhagya, D.; Prema, L.; Rajamohan, T. Therapeutic effects of tender coconut water on oxidative stress in fructose fed insulin resistant hypertensive rats. Asian Pac. J. Trop. Med. 2012, 5, 270–276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gandhi, M.; Aggarwal, M.; Puri, S.; Singla, S.K. Prophylactic effect of coconut water (Cocos nucifera L.) on ethylene glycol induced nephrocalcinosis in male wistar rat. Int. Braz. J. Urol. 2013, 39, 108–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loki, A.; Rajamohan, T. Hepatoprotective and antioxidant effect of tender coconut water on carbon tetrachloride induced liver injury in rats. Indian J. Biochem. Biophys. 2003, 40, 354–357. [Google Scholar]
- Rao, S.; Najam, R. Coconut water of different maturity stages ameliorates inflammatory processes in model of inflammation. J. Intercul. Ethnopharmacol. 2016, 5, 244–249. [Google Scholar] [CrossRef]
- Salil, G.; Nevin, K.G.; Rajamohan, T. Coconut kernel-derived proteins enhance hypolipidemic and antioxidant activity in alloxan-induced diabetic rats. Int. J. Food Sci. Nutr. 2012, 64, 327–332. [Google Scholar] [CrossRef]
- Nguyen, Q.T.; Bandupriya, H.D.D.; Foale, M.; Adkins, S.W. Biology, propagation and utilization of elite coconut varieties (makapuno and aromatics). Plant Physiol. Biochem. 2016, 109, 579–589. [Google Scholar] [CrossRef] [Green Version]
- Luengwilai, K.; Beckles, D.M.; Pluemjit, O.; Siriphanich, J. Postharvest quality and storage life of ‘Makapuno’ coconut (Co-cos nucifera L.). Sci. Hortic. 2014, 175, 105e110. [Google Scholar] [CrossRef] [Green Version]
- Sukendah, H.; Volkaert, H.; Sudarsono, S. Isolation and analysis of DNA fragment of genes related to Kopyor trait in coconut plant. Indones J. Biotechnol. 2009, 14, 1169–1178. [Google Scholar] [CrossRef]
- Carandang, E. Health benefits of virgin coconut oil. Indian Coconut J. Cochin 2008, 38, 8–12. [Google Scholar]
- Tansakul, A.; Chaisawang, P. Thermophysical properties of coconut milk. J. Food Eng. 2006, 73, 276–280. [Google Scholar] [CrossRef]
- Lauzon, R.D. Physico-chemical properties and processing possibilities of macapuno cultivars developed at Leyte State University. Philipp. J. Crop. Sci. 2005, 30, 55e60. [Google Scholar]
- Leorna, M.; Israel, K.A. The influence of maturity of VMAC5 (Cocos nucifera L. ‘makapuno’) on its physicochemical, proxi-mate composition and fatty acid profile. IOP Conf. Ser. Earth Environ. Sci. 2018, 102, 012098. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, B.; Yao, S. High-performance liquid chromatography/electrospray ionization-mass spectrometry for simultaneous determination of taurine and 10 water-soluble vitamins in multivitamin tablets. Anal. Chim. Acta 2006, 569, 169–175. [Google Scholar] [CrossRef]
- DeVries, J.W.; Egberg, D.C.; Heroff, J.C. Concurrent analysis of vitamin A and vitamin E by reversed phase high performance liquid chromatography. In Liquid Chromatographic Analysis of Food and Beverages; Academic Press: Cambridge, MA, USA, 1979; pp. 477–497. [Google Scholar] [CrossRef]
- European Commission. Commission Directive 98/64/EC of 3 September 1998 establishing Community methods of analysis for the determination of amino-acids, crude oils and fats, and olaquindox in feedingstuffs and amending Directive 71/393/EEC. Off. J. Eur. Communities 1998, 257, 14–28. [Google Scholar]
- Çevikkalp, S.A.; Löker, G.B.; Yaman, M.; Amoutzopoulos, B. A simplified HPLC method for determination of tryptophan in some cereals and legumes. Food Chem. 2016, 193, 26–29. [Google Scholar] [CrossRef]
- Attard, E. A Rapid microtitre plate Folin-Ciocalteu method for the assessment of polyphenols. Open Life Sci. 2013, 48, 48–53. [Google Scholar] [CrossRef]
- Chandra, S.; Khan, S.; Avula, B.; Lata, H.; Yang, M.H.; ElSohly, M.A.; Khan, I.A. Assessment of total phenolic and flavonoid content, antioxidant properties, and yield of aeroponically and conventionally grown leafy vegetables and fruit crops: A comparative study. Evid.-Based Complement Altern. Med. 2014, 2014, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Leite, L.; Dourado, L. Laboratory activities, science education and problem-solveing skills. Procedia Soc. Behav. Sci. 2013, 106, 1677–1686. [Google Scholar] [CrossRef] [Green Version]
- Fazel, S.; Hamidreza, M.; Rouhollah, G.; Mohammadreza, V. Spectrophotometric determination of total alkaloids in some Iranian medicinal plants. J. Appl. Hortic. 2010, 12, 69–70. [Google Scholar] [CrossRef]
- Herald, T.; Gadgil, P.; Tilley, M. High-throughput micro plate assays for screening flavonoid content and DPPH-scavenging activity in sorghum bran and flour. J. Sci. Food Agric. 2012, 92, 2326–2331. [Google Scholar] [CrossRef]
- Bolanos de la Torre, A.A.; Henderson, T.; Nigam, P.S.; Owusu-Apenten, R.K. A universally calibrated microplate ferric reducing antioxidant power (FRAP) assay for foods and applications to Manuka honey. Food Chem. 2015, 174, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Byrne, D.H. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food. Compos. Anal. 2010, 19, 669–675. [Google Scholar] [CrossRef]
- Aruoma, O.I. Methodological considerations for characterizing potential antioxidant actions of bioactive components in plant foods. In Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis; Elsevier: Amsterdam, The Netherlands, 2003; Volume 523, pp. 9–20. [Google Scholar] [CrossRef]
- Santoso, U.; Kubo, K.; Ota, T.; Tadokoro, T.; Maekawa, A. Nutrient composition of kopyor coconuts (Cocos nucifera L.). Food Chem. 1996, 57, 299–304. [Google Scholar] [CrossRef]
- Ngampeerapong, C.; Chavasit, V. Nutritional and bioactive compounds in coconut meat of different sources: Thailand, Indonesia and Vietnam. CMU J. Nat Sci. 2019, 18, 562–573. [Google Scholar] [CrossRef]
- United States Department of Agriculture (USDA). FoodData Central. 2019; Nuts, Coconut Meat. Available online: https://fdc.nal.usda.gov/fdc-app.html#/food-details/170169/nutrients/ (accessed on 29 September 2022).
- Trinidad, T.P.; Valdez, D.H.; Loyola, A.S.; Mallillin, A.C.; Askali, F.C.; Castillo, J.C.; Masa, D.B. Glycaemic index of different coconut (Cocos nucifera)-flour products in normal and diabetic subjects. Br. J. Nutr. 2003, 90, 551–556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trinidad, T.P.; Mallillin, A.C.; Valdez, D.H.; Loyola, A.S.; Askali-Mercado, F.C.; Castillo, J.C.; Encabo, R.R.; Masa, D.B.; Maglaya, A.S.; Chua, M.T. Dietary fiber from coconut flour: A functional food. Innov. Food Sci. Emerg. Technol. 2006, 7, 309–317. [Google Scholar] [CrossRef]
- Abbasiliasi, S.; Tan, S.J.; Bello, B.; Ibrahim, T.A.T.; Tam, J.T.; Ariff, A.; Mustafa, S. Prebiotic efficacy of coconut kernel cake’s soluble crude polysaccharides on growth rates and acidifying property of probiotic lactic acid bacteria in vitro. Biotechnol. Biotechnol. Equip. 2019, 33, 1216–1227. [Google Scholar] [CrossRef] [Green Version]
- Hanafi, F.N.A.; Kamaruding, N.A.; Shaharuddin, S. Influence of coconut residue dietary fiber on physicochemical, probiotic (Lactobacillus plantarum ATCC 8014) survivability and sensory attributes of probiotic ice cream. Lebensm. -Wiss. Technol. 2022, 154, 112725. [Google Scholar] [CrossRef]
- Huang, L.; Gao, L.; Chen, C. Role of medium-chain fatty acids in healthy metabolism: A clinical perspective. Trends Endocrinol. Metab. 2021, 32, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Labarthe, F.; Gélinas, R.; Des Rosiers, C. Medium-chain fatty acids as metabolic therapy in cardiac disease. Cardiovasc. Drugs Ther. 2008, 22, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Wang, Y.; Jiang, Y.; Zhang, Z.; Sun, X.; Yu, L.L. Dietary intake of structured lipids with different contents of medium-chain fatty acids on obesity prevention in C57BL/6J mice. J. Food Sci. 2017, 82, 1968–1977. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H. Good fats versus bad fats: A comparison of fatty acids in the promotion of insulin resistance, inflammation, and obesity. Mol. Med. 2017, 114, 303–307. [Google Scholar]
- Budreviciute, A.; Damiati, S.; Sabir, D.K.; Onder, K.; Schuller-Goetzburg, P.; Plakys, G.; Katileviciute, A.; Khoja, S.; Kodzius, R. Management and prevention strategies for non-communicable diseases (NCDs) and their risk factors. Front. Public Health 2020, 8, 574111. [Google Scholar] [CrossRef] [PubMed]
- Tanqueco, R.E.; Rodriguez, F.M.; Laude, R.P.; Cueno, M.E. Total free sugars, oil and total phenolics content of stored coconut (Cocos nucifera L.) water. Philipp. J. Sci. 2007, 136, 103–108. [Google Scholar]
- Tan, T.-C.; Cheng, L.-H.; Bhat, R.; Rusul, G.; Easa, A.M. Composition, physicochemical properties and thermal inactivation kinetics of polyphenol oxidase and peroxidase from coconut (Cocos nucifera) water obtained from immature, mature and overly-mature coconut. Food Chem. 2014, 142, 121–128. [Google Scholar] [CrossRef]
- Mahmood, T.; Anwar, F.; Abbas, M.; Saari, N. Effect of maturity on phenolics (phenolic acids and flavonoids) profile of strawberry cultivars and mulberry species from Pakistan. Int. J. Mol. Sci. 2012, 13, 4591–4607. [Google Scholar] [CrossRef] [Green Version]
- Grosso, G. Dietary antioxidants and prevention of non-communicable diseases. Antioxidants 2018, 7, 94. [Google Scholar] [CrossRef] [Green Version]
- Setiawan, B.; Azra, J.M.; Nasution, Z.; Sulaeman, A.; Estuningsih, S. Development of freeze-dried coconut drink and its nutrient content, sensory profile, and shelf life. J. Culin Sci. Technol. 2022. [Google Scholar] [CrossRef]
- Shen, N.; Wang, T.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem. 2022, 383, 132531. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A.; Cimpeanu, C.; Predoi, G. Antioxidant capacity determination in plants and plant-derived products: A review. Oxid. Med. Cell Longev. 2016, 2016, 9130976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deen, A.; Visvanathan, R.; Wickramarachchi, D.; Marikkar, N.; Nammi, S.; Jayawardana, B.C.; Liyanage, R. Chemical composition and health benefits of coconut oil: An overview. J. Sci. Food Agric. 2021, 101, 2182–2193. [Google Scholar] [CrossRef] [PubMed]
- Karunasiri, A.N.; Gunawardane, M.; Senanayake, C.M.; Jayathilaka, N.; Seneviratne, K.N. Antioxidant and nutritional properties of domestic and commercial coconut milk preparations. Int. J. Food Sci. 2020, 2020, 3489605. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, Y.; Zhang, Y.; Xu, J.; Gao, G. Antioxidant activity of coconut (Cocos nucifera L.) protein fractions. Molecules 2018, 23, 707. [Google Scholar] [CrossRef] [Green Version]
- Olatunde, O.O.; Benjakul, S.; Vongkamjan, K.; Amnuaikit, T. Liposomal encapsulated ethanolic coconut husk extract: Antioxidant and antibacterial properties. J. Food Sci. 2019, 84, 3664–3673. [Google Scholar] [CrossRef]
- Asgha, M.T.; Yusof, Y.A.; Mokhtar, M.N.; Ya’acob, M.E.; Mohd, G.H.; Chang, L.S.; Manaf, Y.N. Coconut (Cocos nucifera L.) sap as a potential source of sugar: Antioxidant and nutritional properties. Food Sci. Nutr. 2019, 8, 1777–1787. [Google Scholar] [CrossRef] [Green Version]
- Santos, J.L.; Bispo, V.S.; Filho, A.B.; Pinto, I.F.; Dantas, L.S.; Vasconcelos, D.F.; Abreu, F.F.; Melo, D.A.; Matos, I.A.; Freitas, F.P.; et al. Evaluation of chemical constituents and antioxidant activity of coconut water (Cocus nucifera L.) and caffeic acid in cell culture. An. Acad. Bras. Cienc. 2013, 85, 1235–1247. [Google Scholar] [CrossRef] [Green Version]
- Gulcin, İ. Antioxidants and antioxidant methods: An updated overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef] [Green Version]
- Tiwari, P.M. Role of flavonoids in DNA damage and carcinogenesis prevention. J. Carcinog. Mutagen. 2017, 8, 4. [Google Scholar] [CrossRef]









| Attribute | Makapuno Meat | Makapuno Water | LOD |
|---|---|---|---|
| Macronutrient (g/100 g) | |||
| Ash * | 0.87–0.89 | 0.70–0.79 | - |
| Calories * | 109.87–125.44 | 23.25–28.82 | - |
| Moisture * | 74.57–75.33 | 92.38–93.74 | - |
| Carbohydrate * | 15.80–20.07 | 4.70–6.22 | - |
| Fat * | 2.43–6.04 | 0.21–0.31 | - |
| Protein (%N*6.25) * | 1.65–1.97 | 0.22–0.64 | - |
| Total Dietary Fiber * | 12.43–16.00 | 1.22–2.09 | - |
| Sugar profile (g/100 g) | |||
| Glucose | 0.81–0.88 | 1.04–1.33 | 0.30 |
| Sucrose | 2.04–2.16 | 2.36–2.73 | 0.30 |
| Total sugar | 2.89–3.02 | 3.43–4.06 | 0.30 |
| Vitamin profile (mg/100 g) | |||
| Vitamin B3 * | 0.00475–0.00570 | 0.00709–0.00802 | 0.00030 |
| Vitamin B5 | 0.00783–0.00910 | 0.00740–0.00830 | 0.00030 |
| Vitamin B6 | <0.00090 | <0.00090 | 0.00030 |
| Vitamin C | 0.640–1.587 | 0.61100–0.82900 | 0.070 |
| Amino acid profile (mg/100 g) | |||
| Glutamic acid | 312.36–362.38 | n.d. | 50.00 |
| Arginine | <250 | n.d. | 100.00 |
| Aspartic acid | <200 | n.d. | 100.00 |
| Alanine | <150 | n.d. | 50.00 |
| Glycine | <100 | n.d. | 50.00 |
| Valine | <100 | n.d. | 50.00 |
| Isoleucine | <100 | n.d. | 50.00 |
| Leucine | <100 | n.d. | 50.00 |
| Histidine | <100 | n.d. | 50.00 |
| Lysine | <100 | n.d. | 50.00 |
| Attribute | Makapuno Meat | Mature Coconut Meat | Makapuno Water | Mature Coconut Water | LOD |
|---|---|---|---|---|---|
| Fatty Acid Profile (g/100 g) | |||||
| C4:0 | n.d. | n.d. | n.d. | n.d. | 0.01 |
| C6:0 | 0.02–0.09 | 0.05–0.07 | n.d. | n.d. | 0.01 |
| C8:0 | 0.25–1.22 | 0.65–0.84 | 0.01–0.02 | n.d. | 0.01 |
| C10:0 | 0.21–0.99 | 0.54–0.69 | 0.01–0.02 | n.d. | 0.01 |
| C12:0 | 0.83–8.35 | 4.39–5.96 | 0.08–0.13 | 0.04–0.05 | 0.01 |
| C14:0 | 0.32–3.07 | 1.47–2.38 | 0.03–0.05 | 0.02–0.03 | 0.01 |
| C16:0 | 0.25–1.49 | 0.9–1.23 | 0.02–0.05 | 0.02–0.04 | 0.01 |
| C18:0 | 0.11–0.61 | 0.37–0.47 | 0.01–0.02 | n.d.-0.01 | 0.01 |
| C18:1n9c | 0.15–0.98 | 0.55–0.86 | 0.01–0.03 | n.d. | 0.01 |
| Saturated fat | 1.83–15.88 | 8.56–11.66 | 0.15–0.25 | 0.08–0.13 | 0.01 |
| Unsaturated fat | 0.17–1.1 | 0.61–0.96 | 0.01–0.03 | 0.02–0.04 | 0.01 |
| Monounsaturated fatty acid | 0.15–0.98 | 0.55–0.86 | 0.01–0.03 | 0.02–0.04 | 0.01 |
| Polyunsaturated fatty acid | 0.02–0.12 | 0.01–0.06 | n.d. | n.d. | 0.01 |
| Omega-6 (mg/100 g) | 16.84–115.01 | 55.75–100.37 | n.d. | n.d. | 10.00 |
| Omega-9 (mg/100 g) | 153.92–984.69 | 549.23–856.17 | 14.83–29.20 | 16.82–39.17 | 10.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phonphoem, W.; Sinthuvanich, C.; Aramrak, A.; Sirichiewsakul, S.; Arikit, S.; Yokthongwattana, C. Nutritional Profiles, Phytochemical Analysis, Antioxidant Activity and DNA Damage Protection of Makapuno Derived from Thai Aromatic Coconut. Foods 2022, 11, 3912. https://doi.org/10.3390/foods11233912
Phonphoem W, Sinthuvanich C, Aramrak A, Sirichiewsakul S, Arikit S, Yokthongwattana C. Nutritional Profiles, Phytochemical Analysis, Antioxidant Activity and DNA Damage Protection of Makapuno Derived from Thai Aromatic Coconut. Foods. 2022; 11(23):3912. https://doi.org/10.3390/foods11233912
Chicago/Turabian StylePhonphoem, Wannarat, Chomdao Sinthuvanich, Attawan Aramrak, Suteekarn Sirichiewsakul, Siwaret Arikit, and Chotika Yokthongwattana. 2022. "Nutritional Profiles, Phytochemical Analysis, Antioxidant Activity and DNA Damage Protection of Makapuno Derived from Thai Aromatic Coconut" Foods 11, no. 23: 3912. https://doi.org/10.3390/foods11233912
APA StylePhonphoem, W., Sinthuvanich, C., Aramrak, A., Sirichiewsakul, S., Arikit, S., & Yokthongwattana, C. (2022). Nutritional Profiles, Phytochemical Analysis, Antioxidant Activity and DNA Damage Protection of Makapuno Derived from Thai Aromatic Coconut. Foods, 11(23), 3912. https://doi.org/10.3390/foods11233912