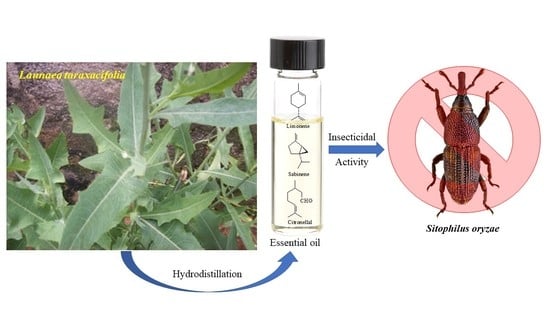
Composition of the Essential Oil and Insecticidal Activity of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey Growing in Nigeria
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Gas Chromatographic–Mass Spectral Analysis
2.3. Insecticidal Activity Screening
3. Results and Discussion
3.1. Essential Oil Composition
3.2. Insecticidal Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ahmed, H. Losses incurred in stored food grains by insect pests—A review. Pak. J. Agric. Res. 1983, 4, 198–207. [Google Scholar]
- Agoda, S.; Atanda, S.; Usanga, O.E.; Ikotun, I.; Isong, I.U. Post-harvest food losses reduction in maize production in Nigeria. Afr. J. Agric. Res. 2011, 6, 4833–4839. [Google Scholar]
- Jones, M.; Alexander, C.; Widmar, N.O.; Ricker-Gilbert, J.; Lowenberg-DeBoer, J.M. Do insect and mold damage affect maize prices in Africa? Evidence from Malawi. Mod. Econ. 2016, 7, 1168–1185. [Google Scholar] [CrossRef] [Green Version]
- Vandekar, M.; Plestina, R.; Wilhelm, K. Toxicity of carbamates for mammals. Bull. World Health Organ. 1971, 44, 241–249. [Google Scholar] [PubMed]
- Hermanutz, R.O. Endrin and malathion toxicity to flagfish (Jordanella floridae). Arch. Environ. Contam. Toxicol. 1978, 7, 159–168. [Google Scholar] [CrossRef]
- Toś-Luty, S.; Obuchowska-Przebirowska, D.; Latuszyńska, J.; Tokarska-Rodak, M.; Haratym-Maj, A. Dermal and oral toxicity of malathion in rats. Ann. Agric. Environ. Med. 2003, 10, 101–106. [Google Scholar] [PubMed]
- Nash, R.G.; Woolson, E.A. Persistence of chlorinated hydrocarbon insecticides in soils. Science 1967, 157, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Talekar, N.S.; Sun, L.-T.; Lee, E.-M.; Chen, J.-S. Persistence of some insecticides in subtropical soil. J. Agric. Food Chem. 1977, 25, 348–352. [Google Scholar] [CrossRef]
- Al-Makkawy, H.K.; Madbouly, M.D. Persistence and accumulation of some organic insecticides in Nile water and fish. Resour. Conserv. Recycl. 1999, 27, 105–115. [Google Scholar] [CrossRef]
- Bondarenko, S.; Gan, J.; Haver, D.L.; Kabashima, J.N. Persistence of selected organophosphate and carbamate insecticides in waters from a coastal watershed. Environ. Toxicol. Chem. 2004, 23, 2649–2654. [Google Scholar] [CrossRef]
- Haubruge, E.; Arnaud, L. Fitness consequences of malathion-specific resistance in red flour beetle (Coleoptera: Tenebrionidae) and selection for resistance in the absence of malathion. J. Econ. Entomol. 2009, 94, 552–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribeiro, B.M.; Guedes, R.N.C.; Oliveira, E.E.; Santos, J.P. Insecticide resistance and synergism in Brazilian populations of Sitophilus zeamais (Coleoptera: Curculionidae). J. Stored Prod. Res. 2002, 39, 21–31. [Google Scholar] [CrossRef]
- Attia, M.A.; Wahba, T.F.; Shaarawy, N.; Moustafa, F.I.; Guedes, R.N.C.; Dewer, Y. Stored grain pest prevalence and insecticide resistance in Egyptian populations of the red flour beetle Tribolium castaneum (Herbst) and the rice weevil Sitophilus oryzae (L.). J. Stored Prod. Res. 2020, 87, 101611. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Koul, O.; Walia, S.; Dhaliwal, G. Essential oils as green pesticides: Potential and constraints. Biopestic. Int. 2008, 4, 63–84. [Google Scholar]
- Reis, S.L.; Mantello, A.G.; Macedo, J.M.; Gelfuso, E.A.; Da Silva, C.P.; Fachin, A.L.; Cardoso, A.M.; Beleboni, R.O. Typical monoterpenes as insecticides and repellents against stored grain pests. Molecules 2016, 21, 258. [Google Scholar] [CrossRef] [Green Version]
- Campolo, O.; Giunti, G.; Russo, A.; Palmeri, V.; Zappalà, L. Essential oils in stored product insect pest control. J. Food Qual. 2018, 2018. [Google Scholar] [CrossRef] [Green Version]
- Burkill, H.M. The Useful Plants of West. Tropical Africa; Volume 1: Families A-D; Royal Botanic Gardens: Kew, UK, 1985. [Google Scholar]
- Cronquist, A. An Integrated System of Classification of Flowering Plants; Columbia University Press: New York, NY, USA, 1981. [Google Scholar]
- Adebisi, A.A. Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey. In Plant Resources of Tropical Africa; Grubben, G.J.H., Denton, O.A., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2004; Volume 2, pp. 103–264. [Google Scholar]
- Adetutu, A.; Olorunnisola, O.S.; Owoade, A.O.; Adegbola, P. Inhibition of In Vivo growth of Plasmodium berghei by Launaea taraxacifolia and Amaranthus viridis in mice. Malar. Res. Treat. 2016, 2016. [Google Scholar] [CrossRef] [Green Version]
- Koukoui, O.; Senou, M.; Agbangnan, P.; Seton, S.; Koumayo, F.; Azonbakin, S.; Adjagba, M.; Laleye, A.; Sezan, A. Effective In Vivo cholesterol and triglycerides lowering activities of hydroethanolic extract of Launaea taraxacifolia leaves. Int. J. Pharm. Sci. Res. 2017, 8, 2040. [Google Scholar]
- Borokini, F.B.; Labunmi, L. In Vitro investigation of antioxidant activities of Launea taraxacifolia and Crassocephalum rubens. Int. J. Food Stud. 2017, 6, 82–94. [Google Scholar] [CrossRef]
- Owoeye, O.; Arinola, G.O. A vegetable, Launaea taraxacifolia, mitigated mercuric chloride alteration of the microanatomy of rat brain. J. Diet. Suppl. 2017, 14, 613–625. [Google Scholar] [CrossRef]
- Adinortey, M.B.; Sarfo, J.K.; Quayson, E.T.; Weremfo, A.; Adinortey, C.A.; Ekloh, W.; Ocran, J. Phytochemical screening, proximate and mineral composition of Launaea taraxacifolia leaves. Res. J. Med. Plants 2012, 6, 171–191. [Google Scholar] [CrossRef] [Green Version]
- Dickson, R.A.; Annan, K.; Fleischer, T.C.; Amponsah, I.K.; Nsiah, K.; Oteng, J.A. Phytochemical investigations and nutritive potential of eight selected plants from Ghana. J. Pharm. Nutr. Sci. 2012, 2, 172–177. [Google Scholar] [CrossRef] [Green Version]
- Adinortey, M.B.; Sarfo, J.K.; Kwarteng, J.; Adinortey, C.A.; Ekloh, W.; Kuatsienu, L.E.; Nyarko, A.K. The ethnopharmacological and nutraceutical relevance of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey. Evidence-Based Complement. Altern. Med. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Bello, O.M.; Abiodun, O.B.; Oguntoye, S.O. Insight into the ethnopharmacology, phytochemistry, pharmacology of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey as an underutilized vegetable from Nigeria: A review. Ann. Univ. Dunarea Jos Galati 2018, 42, 137–152. [Google Scholar]
- Arawande, J.O.; Amoo, I.A.; Lajide, L. Chemical and phytochemical composition of wild lettuce (Launaea taraxacifolia). J. Appl. Phytotechnol. Environ. Sanit. 2013, 2, 25–30. [Google Scholar]
- Dansi, A.; Vodouhè, R.; Azokpota, P.; Yedomonhan, H.; Assogba, P.; Adjatin, A.; Loko, Y.L.; Dossou-Aminon, I.; Akpagana, K. Diversity of the neglected and underutilized crop species of importance in Benin. Sci. World J. 2012, 2012. [Google Scholar] [CrossRef] [Green Version]
- Gbadamosi, I.T.; Okolosi, O. Botanical galactogogues: Nutritional values and therapeutic potentials. J. Appl. Biosci. 2013, 61, 4460–4469. [Google Scholar] [CrossRef] [Green Version]
- Olugbenga, D.J.; Ukpanukpong, R.U.; Ngozi, U.R. Phytochemical screening, proximate analysis and acute toxicity study of Launaea taraxacifolia ethanolic extract on albino rats. Int. J. Sci. Technoledge 2015, 3, 199–202. [Google Scholar]
- Koukoui, O.; Agbangnan, P.; Boucherie, S.; Yovo, M.; Nusse, O.; Combettes, L.; Sohounhloué, D. Phytochemical study and evaluation of cytotoxicity, antioxidant and hypolipidemic properties of Launaea taraxacifolia leaves extracts on cell lines HepG2 and PLB985. Am. J. Plant Sci. 2015, 6, 1768–1779. [Google Scholar] [CrossRef] [Green Version]
- Ruffina, A.N.; Maureen, C.O.; Esther, A.E.; Chisom, I.F. Phytochemical analysis and antibacterial activity of Launaea taraxacifolia ethanolic leave extract. Sch. Acad. J. Biosci. 2016, 4, 193–196. [Google Scholar]
- British Pharmacopoeia; H. M. Starionery Office: London, UK, 1993; Volume I.
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Mondello, L. FFNSC 3; Shimadzu Scientific Instruments: Columbia, MD, USA, 2016. [Google Scholar]
- Satyal, P. Development of GC-MS Database of Essential Oil Components by the Analysis of Natural Essential Oils and Synthetic Compounds and Discovery of Biologically Active Novel Chemotypes in Essential Oils. Ph.D. Thesis, University of Alabama in Huntsville, Huntsville, AL, USA, 2015. [Google Scholar]
- NIST17; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2017.
- Ilboudo, Z.; Dabiré, L.C.B.; Nébié, R.C.H.; Dicko, I.O.; Dugravot, S.; Cortesero, A.M.; Sanon, A. Biological activity and persistence of four essential oils towards the main pest of stored cowpeas, Callosobruchus maculatus (F.) (Coleoptera: Bruchidae). J. Stored Prod. Res. 2010, 46, 124–128. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Finney, D. Probit Analysis, reissue ed.; Cambridge University Press: Cambridge, UK, 2009; ISBN 978-0521135900. [Google Scholar]
- Ololade, Z.S.; Kuyooro, S.E.; Ogunmola, O.O.; Abiona, O.O. Phytochemical, antioxidant, anti-arthritic, anti-inflammatory and bactericidal potentials of the leaf extract of Lactuca teraxacifolia. Glob. J. Med. Res. B Pharma, Drug Discov. Toxicol. Med. 2017, 17, 18–28. [Google Scholar]
- Park, I.K.; Lee, S.G.; Choi, D.H.; Park, J.D.; Ahn, Y.J. Insecticidal activities of constituents identified in the essential oil from leaves of Chamaecyparis obtusa against Callosobruchus chinensis (L.) and Sitophilus oryzae (L.). J. Stored Prod. Res. 2003, 39, 375–384. [Google Scholar] [CrossRef]
- Wang, D.C.; Qiu, D.R.; Shi, L.N.; Pan, H.Y.; Li, Y.W.; Sun, J.Z.; Xue, Y.J.; Wei, D.S.; Li, X.; Zhang, Y.M.; et al. Identification of insecticidal constituents of the essential oils of Dahlia pinnata Cav. against Sitophilus zeamais and Sitophilus oryzae. Nat. Prod. Res. 2015, 29, 1748–1751. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.-H.; Choi, W.-S.; Lee, S.-E.; Park, B.-S. Fumigant toxicity of essential oils and their constituent compounds towards the rice weevil, Sitophilus oryzae (L.). Crop Prot. 2001, 20, 317–320. [Google Scholar] [CrossRef]
- García, M.; Donadel, O.J.; Ardanaz, C.E.; Tonn, C.E.; Sosa, M.E. Toxic and repellent effects of Baccharis salicifolia essential oil on Tribolium castaneum. Pest Manag. Sci. 2005, 61, 612–618. [Google Scholar] [CrossRef]
- Liu, T.-T.; Chao, L.K.-P.; Hong, K.-S.; Huang, Y.-J.; Yang, T.-S. Composition and insecticidal activity of essential oil of Bacopa caroliniana and interactive effects of individual compounds on the activity. Insects 2020, 11, 23. [Google Scholar] [CrossRef] [Green Version]
- Samarasekera, R.; Kalkari, K.S.; Weerasinghe, I.S. Insecticidal activity of essential oils of Ceylon Cinnamomum and Cymbopogon species against Musca domestica. J. Essent. Oil Res. 2006, 18, 352–354. [Google Scholar] [CrossRef]
- Saad, M.M.G.; Abou-Taleb, H.K.; Abdelgaleil, S.A.M. Insecticidal activities of monoterpenes and phenylpropenes against Sitophilus oryzae and their inhibitory effects on acetylcholinesterase and adenosine triphosphatases. Appl. Entomol. Zool. 2018, 53, 173–181. [Google Scholar] [CrossRef]
- Bossou, A.D.; Ahoussi, E.; Ruysbergh, E.; Adams, A.; Smagghe, G.; De Kimpe, N.; Avlessi, F.; Sohounhloue, D.C.K.; Mangelinckx, S. Characterization of volatile compounds from three Cymbopogon species and Eucalyptus citriodora from Benin and their insecticidal activities against Tribolium castaneum. Ind. Crops Prod. 2015, 76, 306–317. [Google Scholar] [CrossRef]
- Yildirim, E.; Emsen, B.; Kordali, S. Insecticidal effects of monoterpenes on Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae). J. Appl. Bot. Food Qual. 2013, 86, 198–204. [Google Scholar]
- Abdelgaleil, S.A.M.; Mohamed, M.I.E.; Badawy, M.E.I.; El-Arami, S.A.A. Fumigant and contact toxicities of monoterpenes to Sitophilus oryzae (L.) and Tribolium castaneum (Herbst) and their inhibitory effects on acetylcholinesterase activity. J. Chem. Ecol. 2009, 35, 518–525. [Google Scholar] [CrossRef] [PubMed]
| Constituents | RIcalc 1 | RIdb 2 | Relative Abundance (%) |
|---|---|---|---|
| α-Pinene | 941 | 933 [37] | 0.9 |
| Sabinene | 976 | 971 [37] | 18.8 |
| Myrcene | 993 | 991 [37] | 2.2 |
| α-Terpinene | 1018 | 1018 [37] | 0.6 |
| Limonene | 1032 | 1030 [37] | 48.8 |
| (Z)-β-ocimene | 1042 | 1034 [37] | 0.9 |
| (E)-β-ocimene | 1052 | 1045 [37] | 4.6 |
| γ-Terpinene | 1062 | 1058 [37] | 1.0 |
| Terpinolene | 1088 | 1086 [36] | 0.4 |
| Linalool | 1101 | 1099 [38] | 3.1 |
| Citronellal | 1155 | 1151 [38] | 11.0 |
| Terpinen-4-ol | 1178 | 1180 [37] | 1.4 |
| 1-Dodecene | 1192 | 1192 [39] | 0.5 |
| n-Dodecane | 1200 | 1200 [36] | 0.5 |
| Neryl acetate | 1366 | 1366 [39] | 0.7 |
| 1-Tetradecene | 1392 | 1388 [36] | 0.5 |
| n-Tetradecane | 1400 | 1400 [36] | 0.2 |
| β-Caryophyllene | 1420 | 1417 [36] | 1.5 |
| α-Humulene | 1456 | 1452 [36] | 0.1 |
| Bicyclogermacrene | 1495 | 1497 [38] | 0.3 |
| Germacrene B | 1556 | 1559 [36] | 0.2 |
| Caryophyllene oxide | 1581 | 1582 [36] | 0.3 |
| 1-Hexadecene | 1592 | 1588 [36] | 0.7 |
| Pentadecanal | 1712 | 1715 [38] | 1.0 |
| Monoterpene hydrocarbons | 78.1 | ||
| Oxygenated monoterpenoids | 16.2 | ||
| Sesquiterpene hydrocarbons | 2.1 | ||
| Oxygenated sesquiterpenoids | 0.3 | ||
| Non-terpene derivatives | 3.3 | ||
| Total identified (%) | 100 |
| Mean % Mortality (±SE) 1 | |||||
|---|---|---|---|---|---|
| Concentration (µL/mL) | 24 h | 48 h | 72 h | 96 h | 120 h |
| 10.00 | 7.50 ± 5.00 cd | 25.00 ± 12.91 cd | 25.00 ± 12.91 de | 25.00 ± 12.91 c | 52.50 ± 17.08 c |
| 20.00 | 15.00 ± 5.77 cd | 30.00 ± 14.14 c | 47.50 ± 17.08 cd | 60.00 ± 14.14 b | 75.00 ± 5.77 b |
| 30.00 | 22.50 ± 9.57 bc | 42.50 ± 12.58 bc | 57.50 ± 9.57 bc | 75.00 ± 5.77 b | 75.00 ± 5.77 b |
| 40.00 | 45.00 ± 17.32 a | 65.00 ± 12.91 b | 75.00 ± 5.77 b | 97.50 ± 5.00 a | 100.00 ± 0.00 a |
| EtOH control | 2.50 ± 5.00 d | 5.00 ± 5.77 d | 10.00 ± 8.16 e | 12.50 ± 9.57 c | 25.00 ± 5.77 d |
| Permethrin | 40.00 ± 0.00 ab | 100.00 ± 0.00 a | 100.00 ± 0.00 a | 100.00 ± 0.00 a | 100.00 ± 0.00 a |
| F-value, DF 2 | 15.08, 5 | 37.44, 5 | 39.69, 5 | 62.21, 5 | 51.13, 5 |
| Contact Time | |||||
|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | 96 h | 120 h | |
| LC50 (95% confidence limits) | 54.38 (39.26−133.8) | 31.64 (23.86−55.67) | 21.48 (16.62−27.21) | 16.38 (13.56−18.78) | 10.10 (5.67−13.31) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Owolabi, M.S.; Ogundajo, A.L.; Alafia, A.O.; Ajelara, K.O.; Setzer, W.N. Composition of the Essential Oil and Insecticidal Activity of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey Growing in Nigeria. Foods 2020, 9, 914. https://doi.org/10.3390/foods9070914
Owolabi MS, Ogundajo AL, Alafia AO, Ajelara KO, Setzer WN. Composition of the Essential Oil and Insecticidal Activity of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey Growing in Nigeria. Foods. 2020; 9(7):914. https://doi.org/10.3390/foods9070914
Chicago/Turabian StyleOwolabi, Moses S., Akintayo L. Ogundajo, Azeezat O. Alafia, Kafayat O. Ajelara, and William N. Setzer. 2020. "Composition of the Essential Oil and Insecticidal Activity of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey Growing in Nigeria" Foods 9, no. 7: 914. https://doi.org/10.3390/foods9070914
APA StyleOwolabi, M. S., Ogundajo, A. L., Alafia, A. O., Ajelara, K. O., & Setzer, W. N. (2020). Composition of the Essential Oil and Insecticidal Activity of Launaea taraxacifolia (Willd.) Amin ex C. Jeffrey Growing in Nigeria. Foods, 9(7), 914. https://doi.org/10.3390/foods9070914