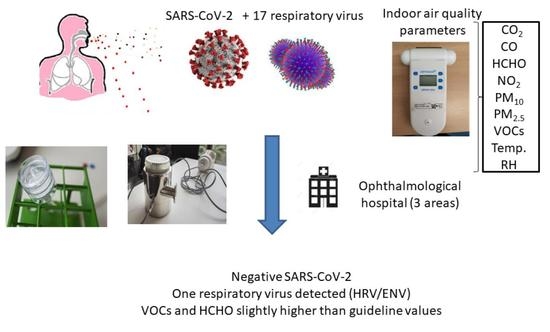
Indoor Air Quality including Respiratory Viruses
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Characterization
2.2. Air Sampling
2.3. Surface Sampling
2.4. Indoor Air Quality (IAQ)
2.5. Sample Preparation for Analyzed Virus
2.6. Virus Analysis
2.6.1. SARS-CoV-2 Detection by RT-PCR
2.6.2. Respiratory Virus Detection by RT-PCR
2.7. Quality Control for Air Sampling and the Virus Detection
3. Results and Discussion
3.1. Previous Sampling Test
3.2. Airborne Viruses and Surface Results
3.3. Indoor Air Quality (IAQ)
4. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Listings of WHO’s Response to COVID-19. 2020. Available online: https://www.who.int/es/news/item/27-04-2020-who-timeline---covid-19 (accessed on 4 October 2020).
- Spanish Ministry of Health. 2021. Consolidated Data at 19 Ferburary 2021. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/Actualizacion_316_COVID-19.pdf (accessed on 16 June 2021).
- Ong, S.W.X.; Tan, Y.K.; Chia, P.Y.; Lee, T.H.; Ng, O.T.; Wong, M.S.Y.; Marimuthu, K. Air, Surface Environmental, and Personal Protective Equipment Contamination by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) From a Symptomatic Patient. JAMA 2020, 323, 1610–1612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef] [PubMed]
- Morawska, L.; Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ. Int. 2020, 139, 105730. [Google Scholar] [CrossRef]
- Alcamí, A.; del Val, M.; Hernán, M.; Latassa, P.; Jiménez, J.L.; Querol, X.; Sánchez, G.; Valencia, A. Informe Científico sobre Vías de Transmisión SARS-CoV-2. Informe para el Ministerio de Ciencia e Innovación de España. 2020. Available online: https://www.ciencia.gob.es/stfls/MICINN/Prensa/FICHEROS/Informe_aerosoles_COVID_MCienciaInnov.pdf (accessed on 30 January 2021).
- Chia, P.Y.; Coleman, K.K.; Tan, Y.K.; Xiang-Ong, S.W.; Gum, M.; Lau, S.K.; Lim, X.F.; Lim, A.S.; Sutijputo, S.; Lee, P.H.; et al. Detection of air and surface contamination by SARS-CoV-2 in hospital rooms of infected patients. Nat. Commun. 2020, 11, 2800. [Google Scholar] [CrossRef] [PubMed]
- Kenarkoohi, A.; Noorimotlagh, Z.; Falahi, S.; Amorloei, A.; Mirzaee, S.A.; Pakzad, I.; Bastani, E. Hospital indoor air quality monitoring for the detection of SARS-CoV-2 (COVID-19) virus. Sci. Total Environ. 2020, 748, 141324. [Google Scholar] [CrossRef]
- Liu, Y.; Ning, Z.; Chen, Y.; Guo, M.; Liu, Y.; Gali, N.K.; Sun, L.; Duan, Y.; Cai, J.; Westerdahl, D.; et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 2020, 582, 557–560. [Google Scholar] [CrossRef]
- Santarpia, J.L.; Rivera, D.N.; Herrera, V.L.; Morwitzer, M.J.; Creager, M.H.; Santarpia, G.W.; Crown, K.K.; Brett-Major, D.M.; Schnaubelt, E.R.; Broadhurst, M.J.; et al. Aerosol and surface contamination of SARS-CoV-2 observed in quarantine and isolation care. Sci. Rep. 2020, 10, 12732. [Google Scholar] [CrossRef]
- Mitchell, A.B.; Tang, B.; Shojaei, M.; Barnes, L.S.; Nalos, M.; Oliver, B.G.; McLean, A.S. A novel sampling method to detect airborne influenza and other respiratory viruses in mechanically ventilated patients: A feasibility study. Ann. Intensive Care 2018, 8, 45–53. [Google Scholar] [CrossRef] [Green Version]
- Blachere, F.M.; Lindsley, W.G.; Pearce, T.A.; Anderson, S.E.; Fisher, M.; Khakko, R.; Meade, B.J.; Lander, O.; Davis, S.; Twelis, R.E.; et al. Measurement of airborne influenza virus in a hospital emergency department. Clin. Infect. Dis. 2009, 48, 438–440. [Google Scholar] [CrossRef] [Green Version]
- Knibbs, L.D.; Morawska, L.; Bell, S.C.; Grzybowski, P. Room ventilation and the risk of airborne infection transmission in 3 health care settings within a large teaching hospital. Am. J. Infect Control 2011, 39, 866–872. [Google Scholar] [CrossRef] [Green Version]
- Rahmani, A.R.; Leili, M.; Azarian, G.; Poormohammadi, A. Sampling and detection of corona viruses in air: A mini review. Sci. Total Environ. 2020, 740, 140207. [Google Scholar] [CrossRef] [PubMed]
- Pena, P.; Morais, J.; Gomes, A.Q.; Viegas, C. Sampling methods and assays applied in SARS-CoV-2 exposure assessment. Sci. Total Environ. 2021, 775, 145903. [Google Scholar] [CrossRef]
- Villanueva, F.; Notario, A.; Cabañas, B.; Martín, P.; Salgado, S.; Gabriel, M.F. Assessment of CO2 and aerosol (PM2.5, PM10, UFP) concentrations during the reopening of schools in the COVID-19 pandemic: The case of a metropolitan area in Central-Southern Spain. Environ. Res. 2021, 197, 111092. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Lednicky, J.A.; Wu, C.Y. Collection, particle sizing and detection of airborne viruses. J. Appl. Microbiol. 2019, 127, 1596–1611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, J.; Bose, M.E.; Beck, E.T.; Fan, J.; Tiwari, S.; Metallo, J.; Jurgens, L.A.; Kehl, S.C.; Ledeboer, N.; Kumar, S.; et al. Rapid Multiplex Reverse Transcription-PCR Typing of Influenza A and B virus, and subtyping of Influenza A virus into H1,2,3,5,7,9, N1 (Human), N1 (Animal), N2, and N7, Including Typing of Novel Swine Origin Influenza A (H1N1) Virus, during the 2009 Outbreak in Milwaukee, Wisconsin. J. Clin. Microbiol. 2009, 47, 2772–2778. [Google Scholar] [CrossRef] [Green Version]
- Suwannakarn, K.; Payungporn, S.; Chieochansin, T.; Samransamruajkit, R.; Amonsin, A.; Songserm, T.; Chaisingh, A.; Chamnanpood, P.; Chutinimitkul, S.; Theamboonlers, A.; et al. Typing (A/B) and subtyping (H1/H3/H5) of influenza viruses by multiplex real-time RT-PCR assays. J. Virol. Methods 2008, 152, 25–31. [Google Scholar] [CrossRef]
- Kuypers, J.; Martin, E.T.; Heugel, J.; Wright, N.; Morrow, R.; Englund, J.A. Clinical disease in children associated with newly described coronavirus subtypes. Pediatrics 2007, 119, 70–76. [Google Scholar] [CrossRef]
- Matsuzaki, Y.; Takashita, E.; Okamoto, M.; Mizuta, K.; Itagaki, T.; Katsushima, F.; Katsushima, Y.; Nagai, Y.; Nishimira, H. Evaluation of a new rapid antigen test using immunochromatography for detection of human metapneumovirus in comparison with real-time PCR assay. J. Clin. Microbiol. 2009, 47, 2981–2984. [Google Scholar] [CrossRef] [Green Version]
- Neske, F.; Blessing, K.; Tollmann, F.; Schubert, J.; Rethwilm, A.; Kreth, H.W.; Weissbrich, B. Real-time PCR for diagnosis of human bocavirus infections and phylogenetic analysis. J. Clin. Microbiol. 2007, 45, 2116–2122. [Google Scholar] [CrossRef] [Green Version]
- Van de Pol, A.C.; Van Loon, A.M.; Wolfs, T.F.W.; Jansen, N.J.G.; Nijhuis, M.; Breteler, E.K.; Schuurman, R.; Roosen, J.W.A. Increased detection of respiratory syncytial virus, influenza viruses, parainfluenza viruses, and adenoviruses with real-time PCR in samples from patients with respiratory symptoms. J. Clin. Microbiol. 2007, 45, 2260–2262. [Google Scholar] [CrossRef] [Green Version]
- Tapparel, C.; Junier, T.; Gerlach, D.; Van Belle, S.; Turin, L.; Cordey, S.; Mühlemann, K.; Regamey, N.; Aubert, J.-D.; Soccal, P.M.; et al. New Respiratory Enterovirus and Recombinant Rhinoviruses among Circulating Picornaviruses. Emerg. Infect. Dis. 2009, 15, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; An, S.; Hwang, J. An integrated system of air sampling and simultaneous enrichment for rapid biosensing of airborne coronavirus and influenza virus. Biosens. Biolectron. 2020, 170, 112656. [Google Scholar] [CrossRef] [PubMed]
- Burton, N.C.; Grinshpun, S.A.; Reponen, T. Physical collection efficiency of filter materials for bacteria and viruses. Ann. Occup. Hyg. 2007, 51, 143–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myatt, T.A.; Johnston, S.L.; Zuo, Z.; Wand, M.; Kebadze, T.; Rudnick, S.; Milton, D.K. Detection of Airborne Rhinovirus and Its Relation to Outdoor Air Supply in Office Environments. Am. J. Respir Crit. Care Med. 2003, 169, 1187–1190. [Google Scholar] [CrossRef] [PubMed]
- Booth, T.F.; Kournikakis, B.; Bastien, N.; Ho, J.; Kobasa, D.; Stadnyk, L.; Li, Y.; Spence, M.; Paton, S.; Henry, B. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J. Infect. Dis. 2005, 191, 1472–1477. [Google Scholar] [CrossRef] [PubMed]
- Lindsley, W.G.; Green, B.J.; Blachere, F.M.; Martin, S.B.; Law, B.; Jensen, P.; Schafer, M. Sampling and Characterization of Bioaerosols. NIOSH Manual of Analytical Methods, 5th ed.; National Institute for Occupational Safety and Health: Cincinnati, OH, USA, 2017.
- Kim, S.-H.; Chang, S.Y.; Sung, M.; Park, J.H.; Hong, B.K.; Lee, H.; Choi, J.-P.; Choi, W.S.; Min, J.-Y. Extensive viable Middle East respiratory syndrome (MERS) coronavirus contamination in air and surrounding environment in MERS isolation wards. Rev. Infect. Dis. 2016, 63, 363–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Aarmink, A.; Wang, W.; Fabri, T.; Koerkamp, P.W.G.; de Jong, M.C. Airborne virus sampling-efficiencies of samplers and their detection limits for infectious bursal disease virus (IBDV). Ann. Agric. Environ. Med. 2014, 21, 464–471. [Google Scholar] [CrossRef] [Green Version]
- Faridi, S.; Niazi, S.; Sadeghi, K.; Naddafi, K.; Yavarian, J.; Shamsipour, M.; Jandaghi, N.Z.S.; Sadeghniiat, K.; Nabizadeh, R.; Yunesian, M. A field indoor air measurement of SARS-CoV-2 in the patient rooms of the largest hospital in Iran. Sci. Total Environ. 2020, 725, 138401. [Google Scholar] [CrossRef]
- Choi, J.Y.; Zemke, J.; Philo, S.E.; Bailey, E.S.; Yondon, M.; Gray, G.C. Aerosol Sampling in a Hospital Emergency Room Setting: A complementary Surveillance Method for the Detection of Respiratory Viruses. Front. Public Health. 2018, 6, 174. [Google Scholar] [CrossRef]
- Xie, C.; Lau, E.H.Y.; Yoshida, T.; Yu, H.; Wang, H.; Wu, H.; Wei, J.; Cowling, B.; Peiris, M.; Li, Y.; et al. Detection of Influenza and Other Respiratory Viruses in air sampled from a Univeristy Campus: A longitudinal study. Clin. Infect. Dis. 2020, 70, 850–858. [Google Scholar] [CrossRef]
- Spena, A.; Palombi, L.; Corcione, M.; Carestia, M.; Spena, V.A. On the optimal indoor air conditions for SARS-CoV-2 inactivation. An entalphy-based approach. Int. J. Environ. Res. Public Health 2020, 17, 6083. [Google Scholar] [CrossRef] [PubMed]
- Escudero, D.; Boga, J.A.; Fernández, J.; Forcelledo, L.; Balboa, S.; Albillos, R.; Astola, I.; García-Prieto, E.; Álvarez-Argüelles, M.E.; Martín, G.; et al. SARS-CoV-2 analysis on environmental surfaces collected in an intensive care unit: Keeping Ernest Shackleton’s spirit. Intens. Care Med. Exp. 2020, 8, 68. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.; Rickard, H.; Stevenson, D.; Aranega-Bou, P.; Pitman, J.; Crook, A.; Davies, K.; Spencer, A.; Burton, C.; Easterbrook, L.; et al. Detection of SARS-CoV-2 within the healthcare environment: A multi-centre study conducted during the first wave of the COVID-19 outbreak in England. J. Hosp. Infect. 2021, 108, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Ma, B.; Lai, X.; Han, L.; Cao, P.; Zhang, J.; Fu, J.; Zhou, Q.; Wei, S.; Wang, Z.; et al. Air and surface contamination by SARS-CoV-2 virus in a tertiary hospital in Wuhan, China. Int. J. Infect. Dis. 2020, 99, 3–7. [Google Scholar] [CrossRef]
- Pasquarella, C.; Colucci, M.E.; Bizzarro, A.; Veronesi, L.; Affanni, P.; Meschi, T.; Brianti, E.; Vitali, P.; Albertini, R. Detection of SARS-CoV-2 on hospital surfaces. Acta Biomed. 2020, 91, 76–78. [Google Scholar] [CrossRef]
- WHO. WHO Guidelines for Indoor Air Quality. Selected Pollutants. 2010. Available online: http://www.euro.who.int/__data/assets/pdf_file/0009/128169/e94535.pdf (accessed on 22 February 2021).
- WHO. WHO Global Air Quality Guidelines. 2021. Available online: https://apps.who.int/iris/bitstream/handle/10665/345329/9789240034228-eng.pdf?sequence=1&isAllowed=y (accessed on 13 October 2021).
- INSHT. Límite de Exposición Profesional para Agentes Químicos en España. 2021. Available online: https://www.insst.es/documents/94886/1637405/LEP+2021.pdf/3e65c9ac-0708-f262-4808-2562cc9e0134?t=1620397240887 (accessed on 13 July 2021).
- ASHRAE, American Society of Heating, Refrigerating and Air-Conditioning Engineers. ASHRAE Standard. Ventilation for Acceptable Indoor Air Quality, 1st ed.; ASHRAE, American Society of Heating, Refrigerating and Air-Conditioning Engineers: Washington, DC, USA, 2010; Volume 62, pp. 1–58. [Google Scholar]
- LIFTEC and CSIC. Guía Práctica: Ventilación Natural en las Aulas. 2020. Available online: https://drive.google.com/file/d/1VG03H9UPqsTBBw3qNKNmZ2PtUbfSsc6f/view (accessed on 30 January 2021).
- Marr, L.; Miller, S.; Prather, K.; Haas, C.; Bahnfleth, W.; Corsi, R.; Tang, J.; Herrmann, H.; Pollitt, K.; Ballester, J.; et al. FAQs on Protecting Yourself from Aerosol Transmission. Version: 1.87. 2020. Available online: http://tinyurl.com/faqs-aerosol (accessed on 19 May 2021).
- De Gennaro, G.; Dambruoso, P.R.; Loiotile, A.D.; Gilio, A.; Giungato, P.; Tutino, M.; Marzocca, A.; Mazzone, A.; Palmisani, J.; Porcelli, F. Indoor air quality in schools. Environ. Chem. Lett. 2014, 12, 467–482. [Google Scholar] [CrossRef]
- Pegas, P.N.; Alves, C.A.; Evtyugina, M.G.; Nunes, T.; Cerqueira, M.; Franchi, M.; Pio, C.A.; Almeida, S.M.; Freitas, M.C. Indoor air quality in elementary schools of Lisbon in spring. Environ. Geochem. Health 2011, 33, 455–468. [Google Scholar] [CrossRef]
- German Supreme Health Authorities. Guide Values for Indoor Air: First Update of the German Risk Assessment Procedure (Basic Scheme). Communication from the Ad-hoc Working Group on Indoor Guide Values of the Indoor Air Hygiene Commission and the States’ Supreme Health Authorities. Bundesgesundheitsblatt 2012, 55, 279–290. Available online: https://www.umweltbundesamt.de/sites/default/files/medien/378/dokumente/guide_values_for_indoor_air_basic_scheme.pdf (accessed on 29 March 2021).



| Virus | Hall (Height = 0.8 m) | Waiting Room (Height = 0.7 m) | Doctor’s Consultation (Height = 1 m) |
|---|---|---|---|
| Influenza virus type A | Negative | Negative | Negative |
| Influenza virus type B | Negative | Negative | Negative |
| HuCoV 229E | Negative | Negative | Negative |
| HuCoV NL63 | Negative | Negative | Negative |
| HuCoV OC43 | Negative | Negative | Negative |
| HuCoV HKU1 | Negative | Negative | Negative |
| HMPV-A | Negative | Negative | Negative |
| HMPV-B | Negative | Negative | Negative |
| hBoV | Negative | Negative | Negative |
| Parainfluenza virus 1 | Negative | Negative | Negative |
| Parainfluenza virus 2 | Negative | Negative | Negative |
| Parainfluenza virus 3 | Negative | Negative | Negative |
| Parainfluenza virus 4 | Negative | Negative | Negative |
| AdV | Negative | Negative | Negative |
| RSV-type A | Negative | Negative | Negative |
| RSV-type B | Negative | Negative | Negative |
| HRV/ENV | Negative | Positive | Negative |
| Parameter (Unities) | Range | Guideline Value |
|---|---|---|
| CO (ppm) | From < DL (1) to 0.68 ppm | 7.03 ppm (5)/3.43 ppm (6) |
| CO2 (ppm) | From 526 ppm to 802 ppm | 700 ppm (7) |
| NO2 (ppm) | From 0.031 ppm to 0.059 ppm | 0.5 ppm (8) |
| VOCs (ppm) | From 0.17 ppm to 3.02 ppm | 1.32 ppm (9) |
| PM10 (mg/m3) | From < DL (2) mg/m3 to 0.005 mg/m3 | 0.05 mg/m3 (5)/0.045 mg/m3 (6) |
| PM2.5 (mg/m3) | From < DL (3) mg/m3 to 0.001 mg/m3 | 0.025 mg/m3 (5)/0.015 mg/m3 (6) |
| HCHO (mg/m3) | From < DL (4) mg/m3 to 1.35 mg/m3 | 0.37 mg/m3 (8) |
| Temperature (°C) | From 19.34 °C to 26.53 °C | From 17 °C to 27 °C (8) |
| Humidity (%) | From 41.69 % to 49.89 % | From 30 % to 70 % (8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López, A.; Fuentes, E.; Yusà, V.; López-Labrador, F.X.; Camaró, M.; Peris-Martinez, C.; Llácer, M.; Ortolá, S.; Coscollà, C. Indoor Air Quality including Respiratory Viruses. Toxics 2021, 9, 274. https://doi.org/10.3390/toxics9110274
López A, Fuentes E, Yusà V, López-Labrador FX, Camaró M, Peris-Martinez C, Llácer M, Ortolá S, Coscollà C. Indoor Air Quality including Respiratory Viruses. Toxics. 2021; 9(11):274. https://doi.org/10.3390/toxics9110274
Chicago/Turabian StyleLópez, Antonio, Esther Fuentes, Vicent Yusà, F. Xavier López-Labrador, Marisa Camaró, Cristina Peris-Martinez, Martin Llácer, Susana Ortolá, and Clara Coscollà. 2021. "Indoor Air Quality including Respiratory Viruses" Toxics 9, no. 11: 274. https://doi.org/10.3390/toxics9110274
APA StyleLópez, A., Fuentes, E., Yusà, V., López-Labrador, F. X., Camaró, M., Peris-Martinez, C., Llácer, M., Ortolá, S., & Coscollà, C. (2021). Indoor Air Quality including Respiratory Viruses. Toxics, 9(11), 274. https://doi.org/10.3390/toxics9110274