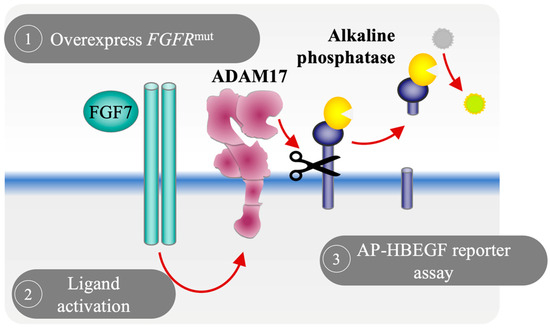
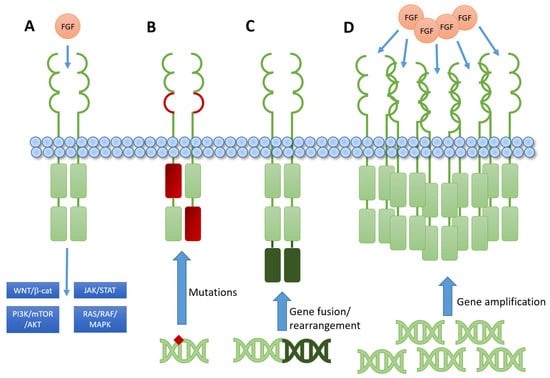
Fibroblast Growth Factor Receptor (FGFR) Signaling Pathway in Tumor 2.0
A topical collection in Cells (ISSN 2073-4409). This collection belongs to the section "Cell Signaling".
Viewed by 6304Editors
Interests: applied and experimental oncology; fibroblast growth factor receptor signaling; telomere maintenance mechanisms; alternative splicing; human and canine tumor cell models; novel therapeutic strategies; biomarkers
Special Issues, Collections and Topics in MDPI journals
Interests: cellular and molecular tumor biology; in vitro models; colon adenomas and carcinomas; growth factor receptor signaling; therapy response, prognostic and predictive markers
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
Following the successful publication of the first Special Issue of “Fibroblast Growth Factor Receptor (FGFR) Signaling Pathway in Tumor”, we are very pleased to launch this new edition, which is dedicated to highlighting the recent translational research in this field, ranging from basic science to clinical studies.
Signaling by fibroblast growth factors (FGFs) and their receptors is an important part of the multilayered network of signal transduction in the cells of the human body, which requires a well-orchestrated interaction to ensure proper functionality. These protein family members of ligands and receptors are crucial for embryonic development and in the adult organism, but are dysregulated in the majority of malignant diseases. Research on FGFRs and related tyrosine receptor kinases in healthy and cancer cells has resulted in several mostly multi-target inhibitors, already in clinical trials or used as cancer drugs. However, the impact of FGFR signaling on the growth, survival and invasiveness of cancer cells and on healthy cells, driving angiogenesis and metastasis in a paracrine manner, are still not completely understood. Further knowledge may lead to the identification of the therapeutic targets and predictive markers needed for specific cancer therapy.
This Special Issue invites reviews and original papers covering translational research on FGFR signaling, with a strong emphasis on the improvement of knowledge for clinical application.
Dr. Klaus Holzmann
Dr. Brigitte Marian
Collection Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cells is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2700 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- FGFR
- downstream signaling
- targeted therapy
- angiogenesis
- tumor cell motility
- tumor microenvironment
- cancer progression
- therapy resistance
- cancer stem cells