The Role of Non-coding RNA in Cancer (Closed)
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Cancer Biomarkers".
Viewed by 94762Editor
Topical Collection Information
Dear Colleagues,
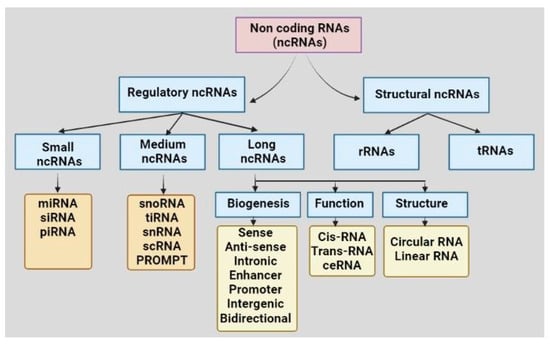
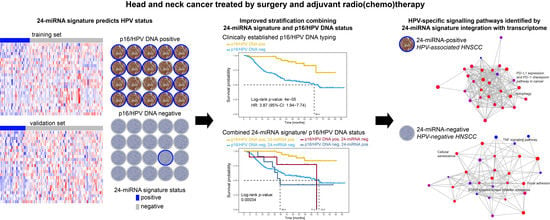
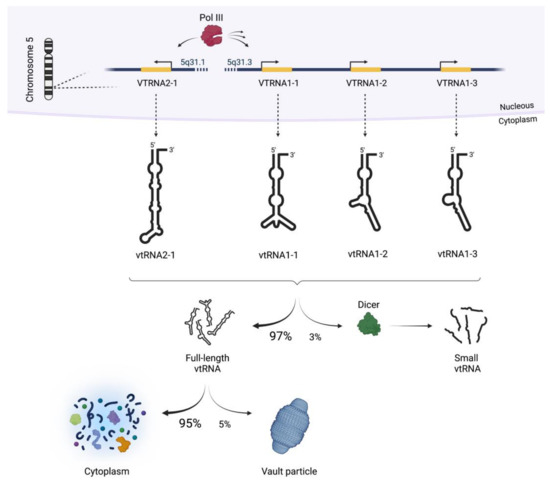
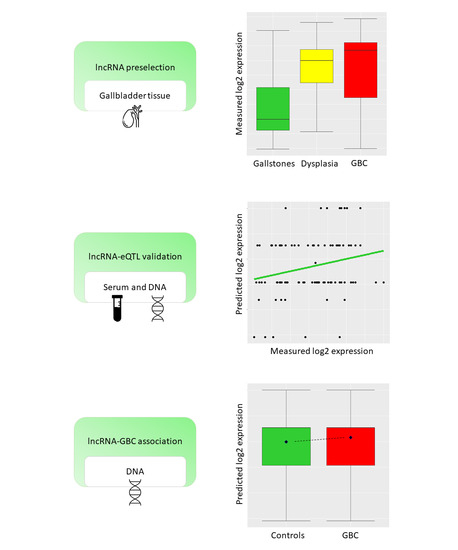
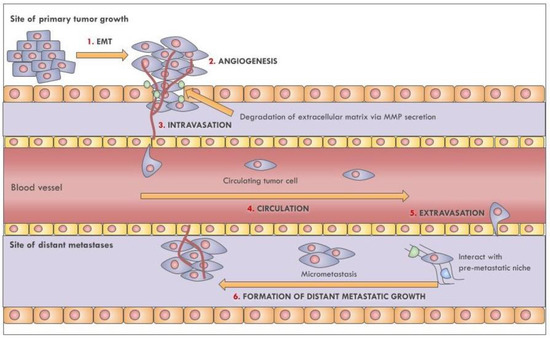
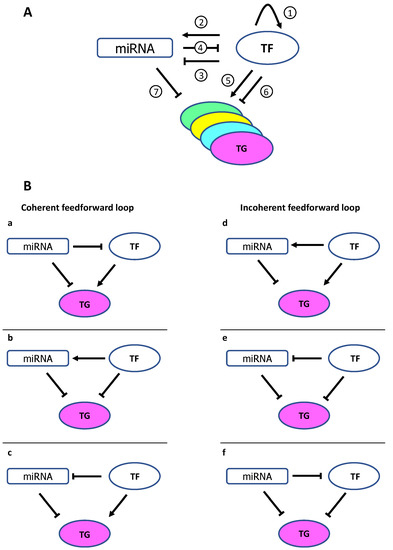
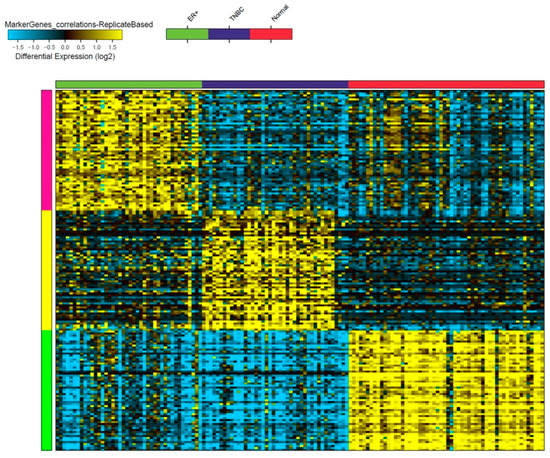
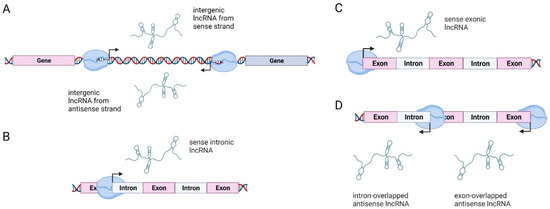
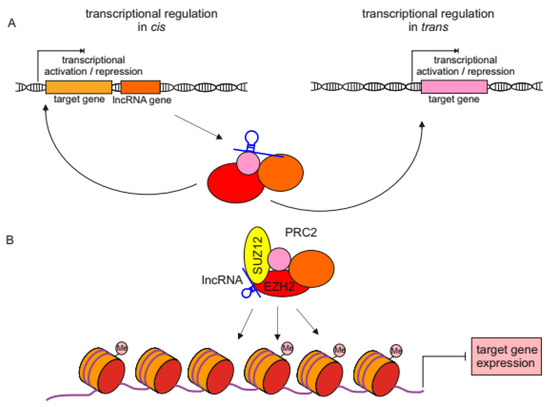
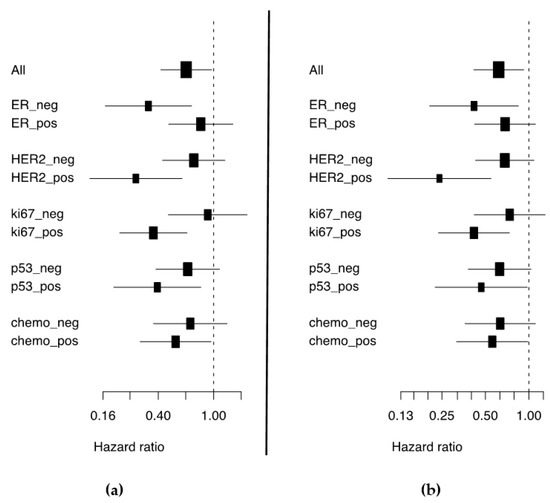
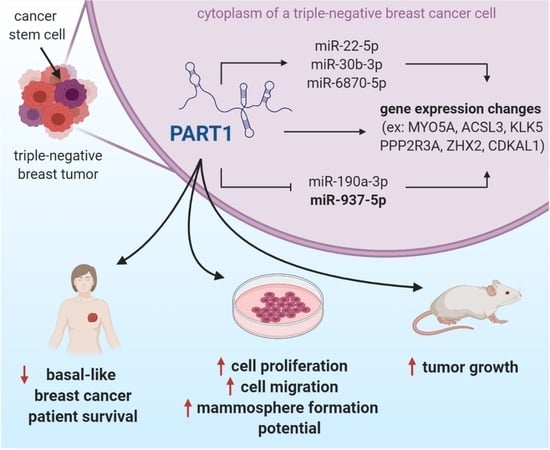
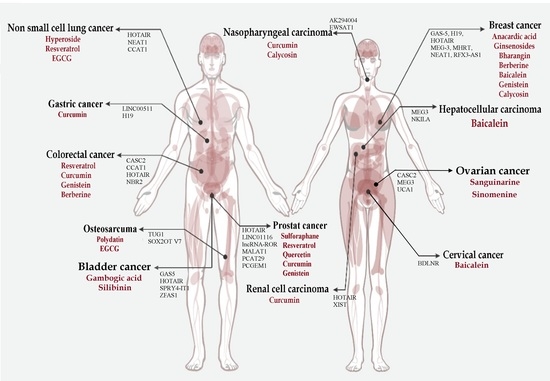
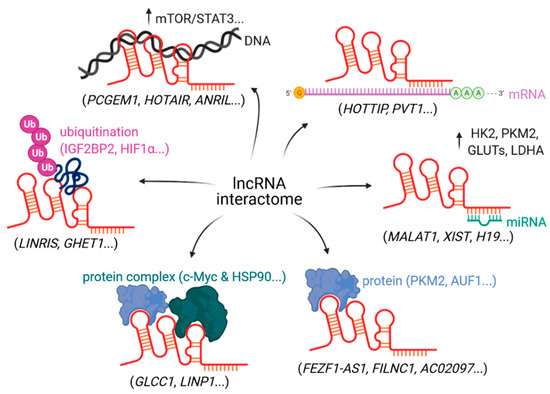
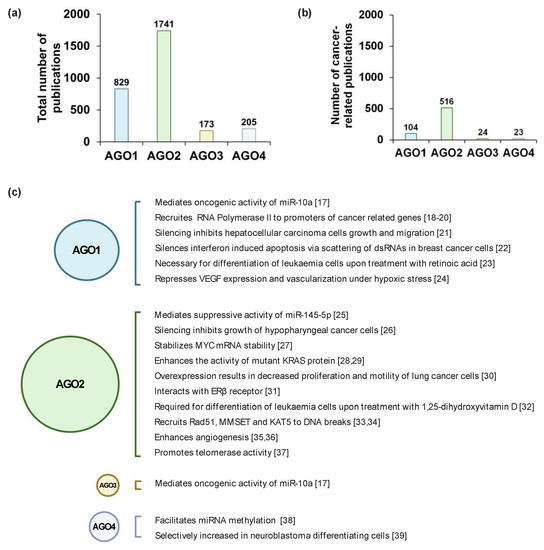
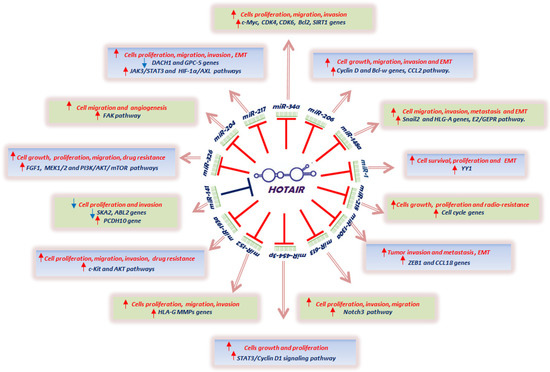
Once considered a byproduct of transcription, the role of non-coding RNA in gene expression is now widely recognized as a critical component of normal development and cell signaling. Just as aberrant gene expression is linked to many disease processes, the dysregulation of ncRNAs, including miRNA and lncRNA, has been associated with cancer initiation and progression. Due to the immense variety of ncRNAs and their mechanisms of action, understanding how altered expression of ncRNA contributes to the disease processes of cancer is a daunting task. Further, the alterations in ncRNA expression levels are often cancer- and even subtype-specific, making broad application of findings challenging. Nonetheless, gaining insight into the nuances of ncRNA regulation offers increasing opportunities for novel therapeutic targets.
In this Topical Collection, we will highlight the complexity of ncRNA-network-regulated gene expression patterns and the implications of aberrant ncRNA in cancer development. We welcome original articles that explore ncRNA as cancer biomarkers, regulators of cell signaling, and potential therapeutic targets. Our goal is to advance our understanding of the mechanisms of ncRNA action and their effects on the signaling networks across malignancies to develop better prognostic and therapeutic strategies.
Dr. Lyndsay Rhodes
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- noncoding RNA
- cancer
- microRNA
- lncRNA
- signaling networks
- targeting